Okay, so you’ve seen it, right? That little squiggle of letters and numbers on a bottle, or maybe scrawled in a lab notebook. FeCl3. It looks like a secret code, doesn't it? Like something a super-spy would use to order a double-shot espresso. But guess what? It's not some clandestine message. It's just… well, it's just the shorthand for something we can all wrap our heads around, even if we haven't touched a chemistry textbook since that one regrettable elective in college.
Think about it like this: you know how when you're texting your bestie, you don't write out "I'll be there in five minutes"? Nope. You hit 'em with "omw" or "cya soon." It's the same principle, really. FeCl3 is just the super-efficient, no-nonsense nickname for a chemical compound. It's the "BRB" of the chemical world, if you will.
But what does this cryptic abbreviation actually mean? Let's break it down, nice and easy. Forget those stuffy professors with their chalk dust and booming lectures. We're going to talk about this like we're chatting over a cup of coffee (or, you know, something stronger). So, buckle up, buttercups, because we're about to demystify FeCl3.
The Big Reveal: What's in a Name?
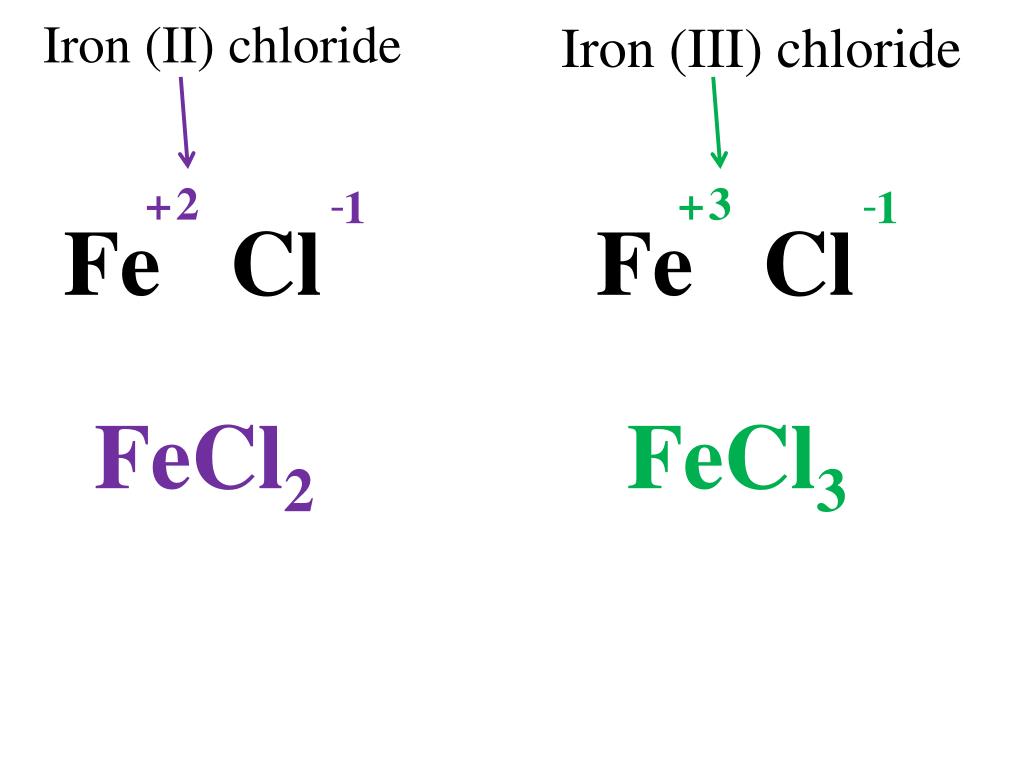
Alright, the moment of truth. That seemingly intimidating FeCl3 actually spells out Iron(III) chloride. Ta-da! Not so scary now, is it? It's like finding out your intimidating boss is actually afraid of fluffy kittens. Suddenly, they seem a lot more approachable.
Let's dissect that a bit. We've got the 'Fe'. If you've ever seen a rusty old bike or a particularly sturdy bridge, you've encountered 'Fe'. It's the symbol for iron. Yep, the very same metal that makes your cast-iron skillet a culinary dream (or a nightmare to scrub, depending on your perspective). Iron is everywhere. It's in your blood, making sure you don't look like a ghost. It's in the earth, giving it that lovely reddish-brown hue in some places. It's the strong, silent type of element.
Then we have the 'Cl3'. Now, 'Cl' is the symbol for chlorine. Chlorine is a bit more… energetic. You know how sometimes you get that weird, slightly antiseptic smell in swimming pools? That's often thanks to chlorine, working its magic to keep things clean and germ-free. It's a bit like that friend who’s always a little too enthusiastic at parties, but ultimately useful.
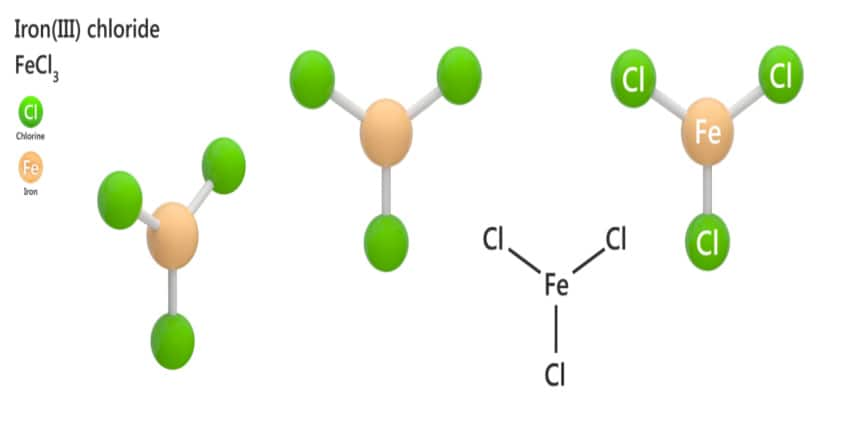
The '3' part? That little guy is telling us how many chlorine atoms are hanging out with our iron atom. It's like saying, "This iron atom has three chlorine buddies with it." They're a little trio, a package deal. Think of it as iron ordering a three-scoop ice cream cone, but instead of sprinkles, it's got chlorine.
Putting It All Together: The "Iron(III)" Conundrum
Now, the ' (III) ' part in Iron(III) chloride is where things get a tiny bit more nuanced, but still totally manageable. You know how sometimes you have a name that's super common, and to tell people apart, you need a middle name or a "Jr." or "Sr."? Well, iron is kind of like that. Iron can exist in different "states," which is a fancy chemistry way of saying it can bond with other elements in slightly different ways, like wearing different hats.
The 'III' is telling us which "hat" our iron is wearing. In this case, it's indicating that the iron atom has lost three of its electrons. Electrons are like tiny, negative charges that orbit around an atom. When an atom loses or gains electrons, it becomes an ion, which is just an atom with an electrical charge. So, Iron(III) is an iron atom that's feeling a little bit lighter, having shed three electrons.

This is crucial because it dictates how iron interacts with other elements. If the iron had lost, say, two electrons, it would be Iron(II), and it would behave differently. It's like having two versions of your favorite superhero – they might have similar powers, but their little quirks make them distinct. So, Iron(III) chloride means we've got our iron atom, feeling a bit zappy with a positive charge, and it's shackled up with three chlorine atoms, which are, in this case, negatively charged.
Why Should You Care About Iron(III) Chloride?
Okay, so you know what it is. But why would you ever need to know this? Well, Iron(III) chloride isn't just sitting around looking pretty in a lab. It's got some real-world applications, some of which might surprise you.
One of the most common uses is in water treatment. Remember how we said chlorine helps keep things clean? Well, Iron(III) chloride acts as a coagulant. Think of it like this: when you're trying to clean a really muddy puddle, sometimes just stirring it up makes it worse. But if you throw in something that makes all the tiny mud particles stick together, they become bigger, heavier clumps that settle to the bottom, leaving you with clearer water. That's basically what Iron(III) chloride does for water. It helps clump up impurities so they can be filtered out.

So, next time you turn on your tap and get a nice, clear glass of water, you can give a silent nod to Iron(III) chloride, doing its dirty work behind the scenes. It’s like the unsung hero of your morning shower.
It's also used in etching. Ever seen those cool circuit boards in electronics? Or intricate metal designs on jewelry? Sometimes, Iron(III) chloride is involved in creating those. It's used to selectively remove material from a metal surface, creating patterns. It's kind of like a very precise, very chemical way of drawing. Imagine having a pen that could literally eat away at metal to create your masterpiece. That's the power of Iron(III) chloride in action!
And then there's its role in medicine. Believe it or not, Iron(III) chloride can be used as an astringent. An astringent is something that causes tissues to contract or shrink. It can be used to stop minor bleeding, like a superhero for your skin. So, if you ever nick yourself shaving and a little dab of something stops the bleed faster than you expected, it might be thanks to a compound that includes Iron(III) chloride.
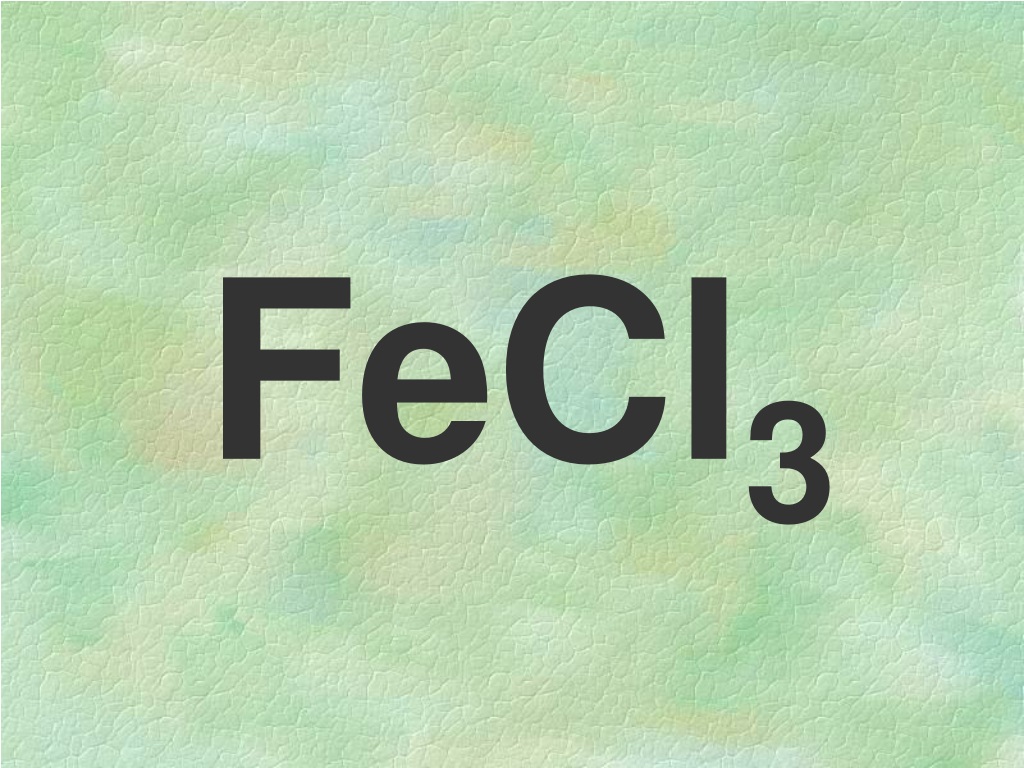
The Chemistry of Everyday Life: It's All Around Us!
See? Chemistry isn't just about beakers and explosions (though, let's be honest, explosions are pretty cool). It's about the stuff that makes our lives easier, cleaner, and more functional. FeCl3, or Iron(III) chloride, is just one tiny example of how the building blocks of the universe are constantly working for us, even when we don't realize it.
It’s like that quiet, reliable friend who always shows up when you need them, never asking for much in return. They just do what needs to be done. Iron(III) chloride is that friend for water purification, for intricate designs, and for a little help when we're a bit worse for wear.
So, the next time you see FeCl3, don't get intimidated. Give it a knowing nod. You know its secret. You know its full name. You know it's not some arcane mystery, but a helpful compound that’s part of the grand, often invisible, tapestry of our everyday lives. It's the chemical equivalent of realizing your neighbor's intimidating poodle is actually a total cuddle bug. Pretty neat, huh?
And if anyone ever asks you what FeCl3 stands for, you can casually drop the bomb: "Oh, that? That's just Iron(III) chloride. You know, for water treatment and stuff." They'll be impressed, and you'll feel like a chemistry whiz, without ever having to memorize the periodic table. You’re welcome!