So, you've been diving into the wild world of covalent bonds with Explore Learning, huh? Awesome! It’s one of those topics that can feel like a puzzle, right? Like, how do these atoms even decide to share their electrons? It’s a whole chemistry party happening at the microscopic level.
And let's be real, sometimes you just get stuck. You stare at a question, you stare at your notes, and your brain just… goes on vacation. Happens to the best of us. So, if you've been Googling "Explore Learning Covalent Bonds answer key," you are so not alone. I get it. We all need a little help sometimes.
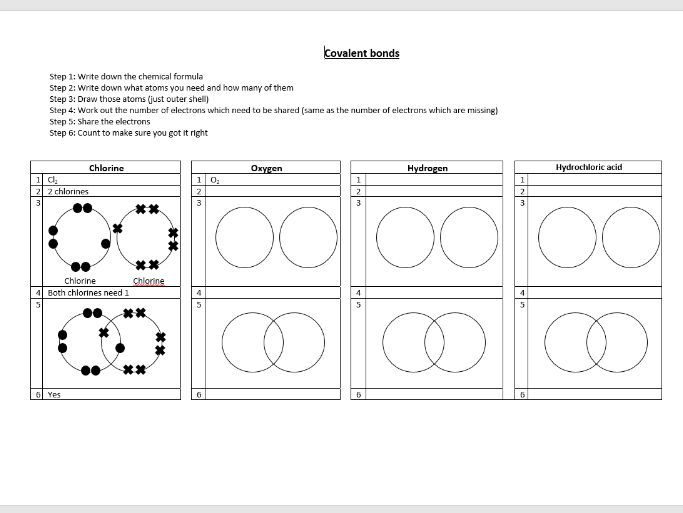
Think of it like this: imagine you and your bestie both have a super cool LEGO brick. But you really need that specific red one to finish your epic spaceship, and your friend also desperately needs it for their race car. What do you do? You probably share, right? "Here, you can have it for a bit, then I'll borrow it!" That, my friend, is basically a covalent bond in action. Atoms doing that electron-sharing dance. So cool, right?
Explore Learning does a pretty neat job of breaking this down, but the "answer key" part… that's a whole other kettle of fish. It's tempting, I know. That little voice whispering, "Just peek! It'll be so easy!" But is it really? Or are we cheating ourselves out of that glorious "aha!" moment? The one where you finally get it, and your brain does a little happy jig? I vote for the jig.
Let's chat about why they share, though. It's all about stability, baby! Atoms are kinda like people at a party. They want to be surrounded by friends, to feel complete. For many atoms, having a full outer electron shell (we're talking the 'valence' electrons, remember?) makes them feel all warm and fuzzy. It's like getting a perfect score on a test – pure satisfaction.
When one atom doesn't quite have enough electrons to get to that happy, full shell, and another atom is in a similar boat, they can team up. They say, "Hey, you know what? We can both be happier if we just… share some of our electrons. Let's form a bond!" And poof! A covalent bond is born. They're essentially saying, "I'll lend you some electrons, and you lend me some electrons, and we'll both pretend they're all ours." Clever, right?
So, when you're wrestling with those Explore Learning questions, think about that sharing. Are the atoms giving electrons away (that's ionic, a different story for another day!) or are they holding hands and sharing? That's the key, pun intended!
Let's talk about the different types of covalent bonds because, surprise, it's not always a perfectly equal split! Sometimes, one atom is a little greedier, I mean, more electronegative. This means it pulls those shared electrons a bit closer to itself. It’s like one friend holding the remote control a little tighter.
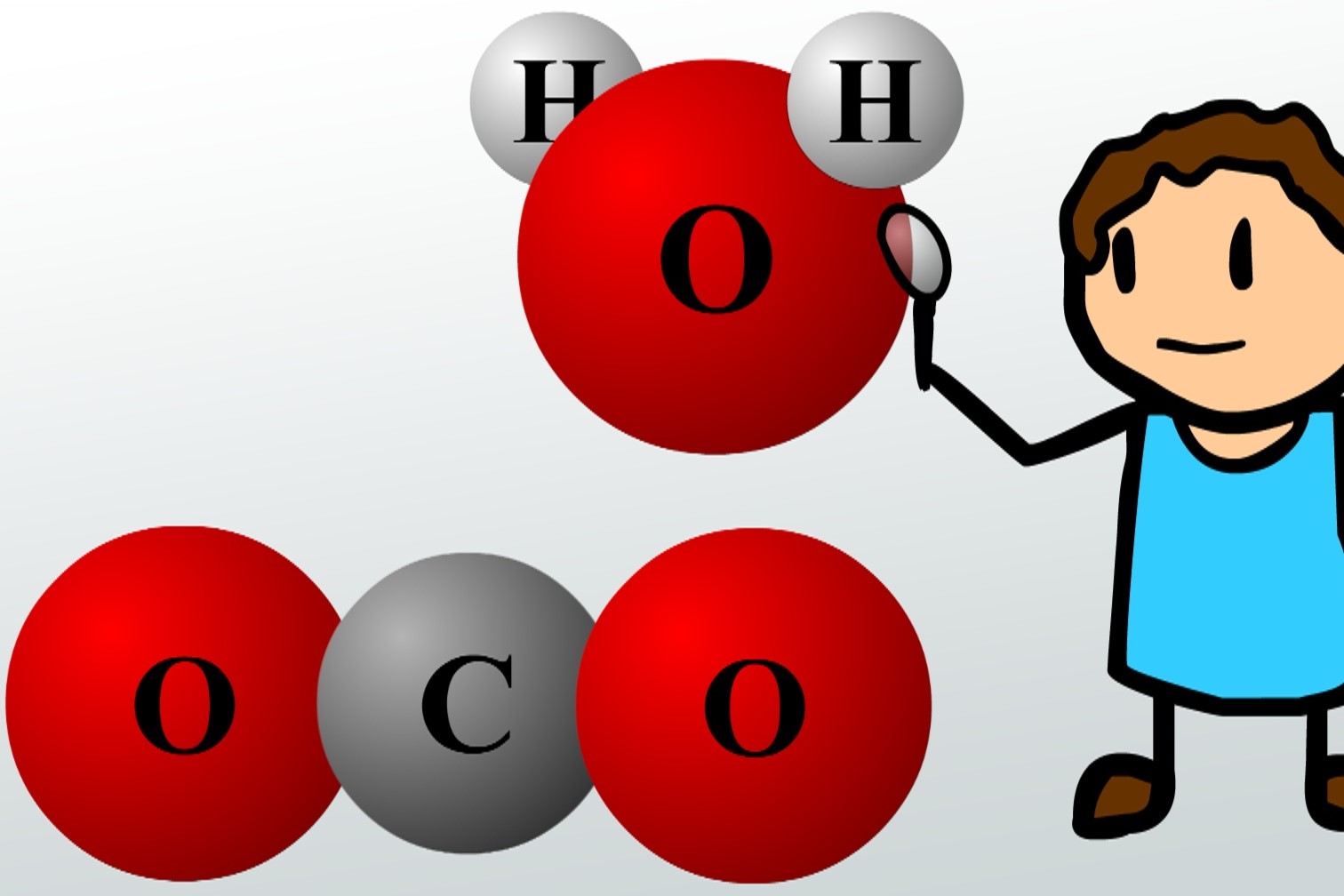
This leads to what we call a polar covalent bond. It's not super uneven, like in ionic bonds where one atom basically steals the electron. No, no. It's more of a gentle tug-of-war. One side of the bond gets a slightly negative charge (because the electrons, which are negative, are hanging out there more) and the other side gets a slightly positive charge. Think of water (H₂O). Oxygen is a bit of a hog with electrons, so the oxygen end of the water molecule is a tad negative, and the hydrogen ends are a tad positive. That's why water is so awesome at dissolving things – those polar molecules are like tiny magnets, attracting all sorts of other things. Mind. Blown.
Then, you have your nonpolar covalent bonds. These are the ultimate BFFs of the electron-sharing world. The electrons are shared equally. It's like two friends splitting a pizza down the middle, no arguments. Atoms with similar electronegativity tend to form these. Think of molecules like oxygen gas (O₂) or methane (CH₄). The electrons are just chilling, equally distributed. No drama, no fuss. Just pure, stable sharing.
When you’re tackling the Explore Learning exercises, pay attention to the atoms involved. Are they wildly different in how much they want electrons? If so, you're probably looking at a polar bond. Are they pretty similar? Nonpolar bond territory! It’s like reading the room, but for atoms.

Now, let's address the elephant in the room, or should I say, the answer key on the internet. It's so tempting, right? You're up late, the caffeine is wearing off, and that last question is just staring at you, mocking you with its blankness. You think, "Just one little peek wouldn't hurt."
But here's the thing. That feeling of accomplishment when you figure it out yourself? It’s gold. It’s way better than just copying an answer. It's like finally solving a tricky riddle – the satisfaction lasts so much longer. And honestly, the Explore Learning platform is designed to help you learn, not just memorize answers. They want you to build that understanding.
If you're struggling, don't despair! That's what the platform is for. It's got explanations, it's got interactive bits, it's got ways to guide you. Maybe you're misunderstanding a fundamental concept. That's okay! That's why you're doing the exercises in the first place. It's a learning process, and sometimes, the learning comes with a few bumps and bruises. Or, you know, a few confused stares at the screen.
So, instead of hunting for an answer key, maybe try a different approach. Revisit the explanations on Explore Learning. Watch a quick YouTube video on covalent bonds (there are SO many good ones out there, and some are even funny!). Talk it through with a classmate. Explaining it to someone else is one of the best ways to solidify your own understanding. Seriously, try it!

And don't forget about Lewis structures! These are like the little stick-figure drawings of molecules that help us visualize those shared electrons. You draw dots for valence electrons, and then you draw lines between atoms to represent the shared pairs. It’s super helpful for seeing how the sharing is happening and whether it’s a single, double, or even a triple bond (woah, triple bonds are intense!).
A single bond is just one pair of shared electrons. Think of two people holding hands. A double bond is two pairs of shared electrons – like two sets of hands holding. And a triple bond? That's three pairs of shared electrons. They're holding on tight! These multiple bonds occur when atoms need to share more electrons to achieve that stable, full outer shell. It's a bit more commitment, you know?
When you're looking at those Lewis structures in your Explore Learning materials, try to predict what kind of bond you'll see. Does the atom need to share two electrons to be happy? Then you might see a double bond! Does it need to share three? Hello, triple bond!
And let’s not forget about the concept of octet rule. This is that general guideline that atoms tend to gain, lose, or share electrons until they are surrounded by eight valence electrons. It’s like the magic number for many atoms to feel complete and stable. Of course, there are exceptions (because science loves to keep us on our toes!), but the octet rule is a really good starting point for understanding why covalent bonds form.

If you're stuck on a particular question, break it down. What atoms are involved? How many valence electrons do they have? What are they trying to achieve? Is it stability? Is it a full outer shell? Once you start asking yourself these questions, the answers start to become clearer. It’s like a detective story for atoms!
Let's say you're looking at a question about methane (CH₄). You know carbon has 4 valence electrons, and hydrogen has 1. Carbon needs 4 more electrons to get to 8. Each hydrogen needs 1 more electron to get to 2 (because hydrogen is cool with just having two, it's a special case, like being the only kid with a juice box at a water-only party). So, the carbon shares one electron with each of the four hydrogens, and each hydrogen shares its one electron with the carbon. Everyone's happy! Four single, nonpolar covalent bonds. See? You can totally do this without an answer key!
The Explore Learning platform is your learning playground. Use all the tools it gives you! Don't just rush through it to get to the end. Take your time. Explore the explanations. Try the practice questions. And if you get something wrong, it’s not a failure. It’s feedback! It’s your chance to learn and grow.
Think about how much more rewarding it will be when you ace that quiz or understand that concept because you worked for it. That feeling of mastery is way more powerful than a temporary fix. Plus, who knows, maybe you'll develop a superpower for understanding chemistry! That's pretty cool, right?
So, put down the imaginary "answer key" and pick up your understanding. Dive deeper into the explanations. Ask yourself those "why" questions. And remember, every covalent bond is just a tiny story of atoms cooperating. It's chemistry at its most fundamental, and it's pretty darn amazing once you start to get it. You've got this!