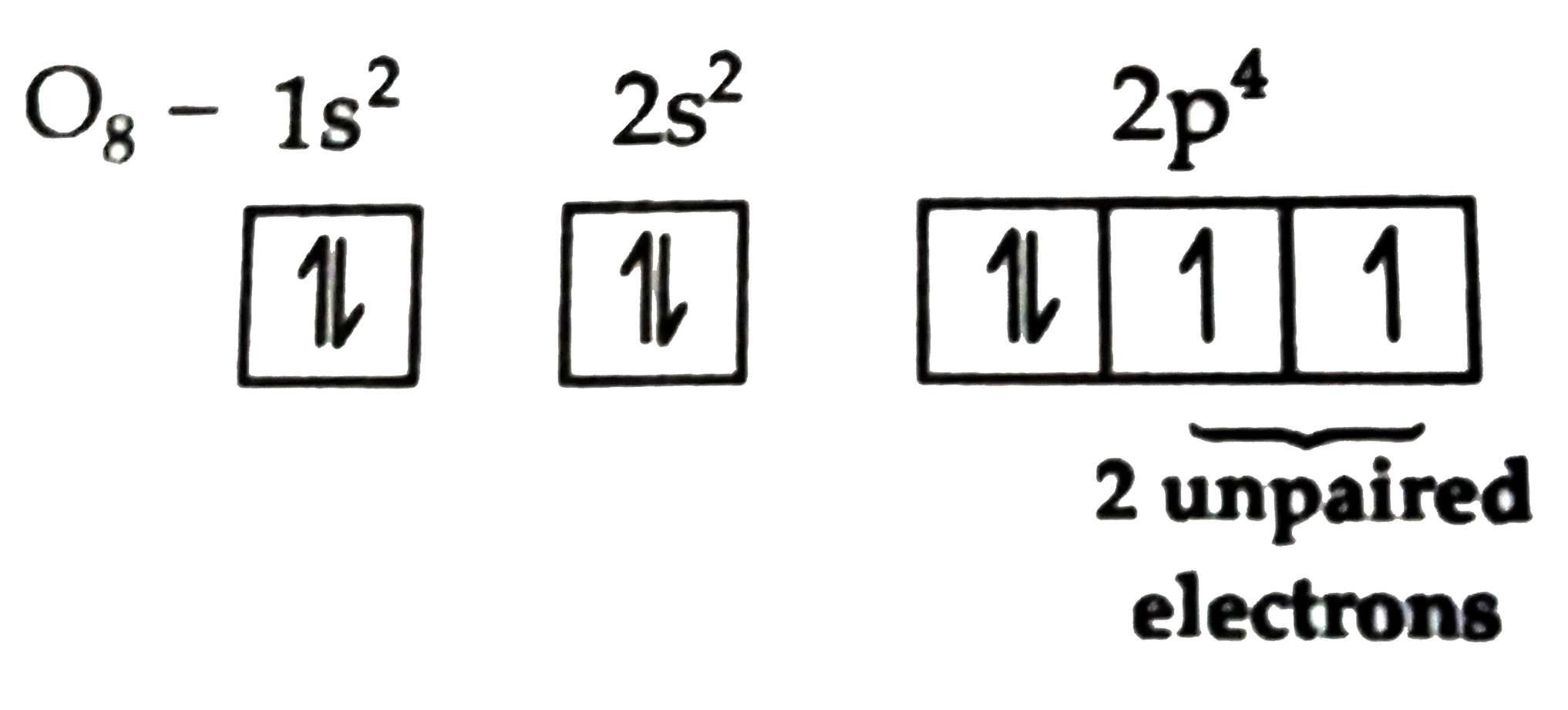
Imagine tiny elemental building blocks, each with its own unique personality. Some are eager to share their toys, others are a bit shy, and then there are those who are just bursting with energy, always ready for a little interaction. Today, we're going to meet a special crew of these elemental buddies: the ones who are just a tiny bit unbalanced, in the most wonderful way.
These aren't your average, perfectly balanced atoms. Think of them like a group of friends who all have one extra bouncy ball they haven't quite figured out what to do with yet. This "extra ball" is what scientists call an unpaired electron. And our special group has not one, not two, but three of these lonely little electrons just hanging out.
It's like having three invisible, energetic siblings in the family. They’re not quite paired up, so they're a bit more restless, a bit more prone to jumping into new games. This makes them incredibly interesting to observe, like watching a playful puppy with too much energy!
These elements are the nitrogen group, a rather distinguished family in the periodic table. We're talking about elements like Nitrogen itself, the very air we breathe, and Phosphorus, famous for its glowing properties and its role in our very bones.
Then there's Arsenic, which has a bit of a dramatic reputation, and Antimony, often found working in alloys, making things stronger. And finally, the less common but equally intriguing Bismuth, known for its beautiful, colorful crystals.
What makes these three unpaired electrons so special? Well, these electrons are like little invitations. They're just itching to find a partner, to form a connection, to join a band!
This eagerness to bond is what gives these elements some of their most amazing properties. They are the quiet organizers of the chemical world, often forming the backbone of important molecules. Think of them as the reliable friends who are always willing to lend a hand, or in this case, an electron.

Let’s start with the most familiar: Nitrogen. This element is everywhere, making up about 78% of the air you’re breathing right now. It’s surprisingly unreactive on its own, much like a sleepy cat. But give it the right conditions, and those three unpaired electrons get to work!
Nitrogen is the essential building block for life. It's in the DNA in every living cell, the very blueprint of who you are. It's in the proteins that build your muscles and enzymes that help you digest your lunch. Pretty important stuff for a gas that seems so lazy!
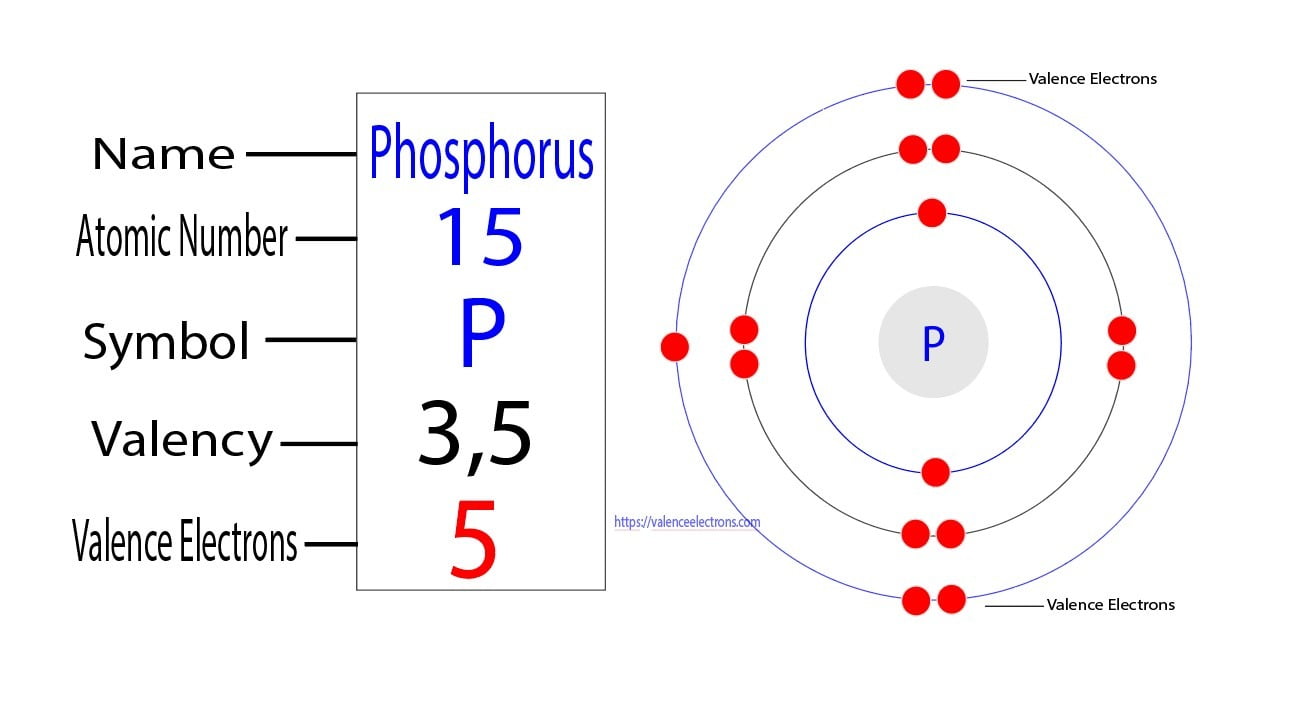
And then there's Phosphorus. This element is a bit of a show-off. It's famous for its ability to glow in the dark, a magical quality that has captivated people for centuries. Imagine discovering a substance that literally lights up the night!
Phosphorus is also crucial for our bodies. It’s a key component of our bones and teeth, giving them strength and structure. It's in the ATP, the energy currency of our cells, powering everything from thinking to running. Without phosphorus, life as we know it would grind to a halt.

Moving on to Arsenic. This element has a bit of a notorious past, often associated with mystery and intrigue. For a long time, its compounds were used as poisons, a dark chapter in its story.
But arsenic isn't all bad! In very small, controlled doses, it can actually be medically important. It's used in some treatments for certain diseases, showing that even seemingly "dangerous" elements have their beneficial sides.
Antimony is the next in line. This element often works behind the scenes, contributing to the strength and durability of other materials. It’s a bit like the quiet but dependable friend who always has your back.
Antimony is used to make things flame-retardant, making our homes and clothes safer. It's also used in batteries, helping to power our modern world. It’s a true team player, making other things better just by being there.

And last but not least, we have Bismuth. This element is the artist of the group. It’s known for forming stunning, iridescent crystals with a rainbow of colors, like a tiny, metallic galaxy.
Bismuth compounds are also used in medicines, particularly for stomach ailments. So, while it looks like something out of a fantasy world, it’s also helping us feel better. A beautiful and beneficial element!
The reason these three elements, and the others in their group, have these three unpaired electrons is a fascinating tale of electron arrangement. It’s like they have three empty "seats" in a special "electron room" that they are eager to fill.
This makes them excellent at forming covalent bonds, where they share their electrons with other atoms. They’re like the ultimate sharers, creating stable molecules by forming strong connections.

Think of it like a party. While some elements might prefer to grab all the snacks for themselves, these nitrogen group elements are happy to share. They're the ones organizing the group games and making sure everyone is included.
This sharing ability is why they are so fundamental to the chemistry of life. They form the stable structures of organic molecules, the complex arrangements that make up everything from a single-celled organism to a redwood tree.
It’s a humbling thought, isn’t it? That these seemingly simple elemental building blocks, with their particular electron personalities, are the architects of our existence. The air we breathe, the food we eat, the very structure of our bodies, all thanks to the industrious nature of these elements with their three unpaired electrons.
So, the next time you take a deep breath, or admire a glowing object, or even just think about the strength of your bones, remember the nitrogen group. Remember those three unpaired electrons, buzzing with potential, eagerly reaching out to form the world around us. They’re not just elements; they’re the quiet, energetic, and essential storytellers of chemistry.
It’s a beautiful reminder that even in the smallest, most fundamental parts of the universe, there’s a dynamic and interconnected dance happening, driven by the simple desire of electrons to find a partner and create something wonderful.