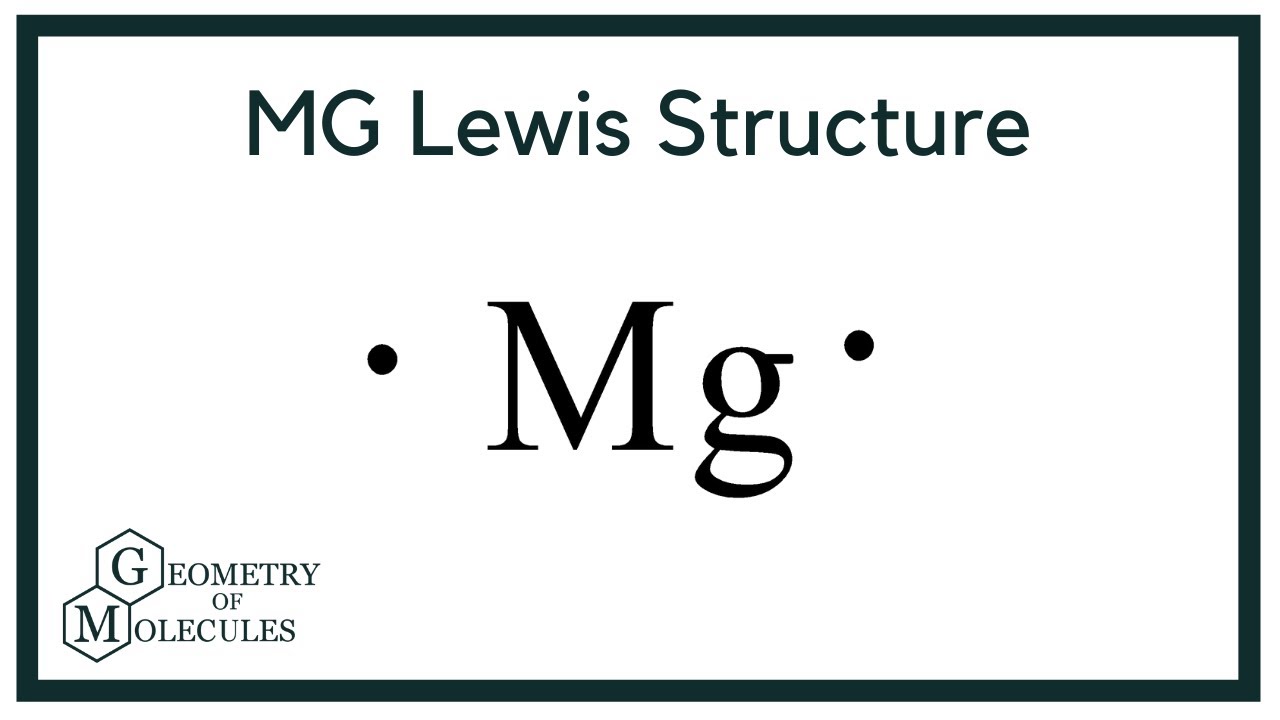
Hey there, coffee buddy! So, we're gonna chat about something super cool today: electron dot structures, specifically for our pal, magnesium oxide. Yeah, I know, "electron dot structure" sounds a bit like a fancy science word, right? But honestly, it's kinda like doodling with atoms. Super simple, once you get the hang of it.
Think of it this way. Atoms are like little LEGO bricks, and they have these tiny things called electrons whizzing around them. These electrons are like the sticky bits that let atoms hold hands, or, you know, bond together. And electron dot structures? They're just a way to draw out those sticky bits so we can see how atoms are gonna get together.
So, magnesium oxide. What even is that? Well, it's actually pretty common. It's what you find in antacids, for goodness sake! You know, those chalky tablets that save your stomach after a spicy burrito. So, we're talking about something important here, people!
Let's break down our two main characters. First up, magnesium. Say it with me: mag-nee-zee-um. It's a metal, which is important. Metals are generally pretty chill, kinda happy to give away their outer electrons. Think of them as generous givers. Magnesium, in particular, is in Group 2 of the periodic table. You know, that little section with the alkali earth metals? That means it's got two electrons hanging out in its outermost shell. Two little guys just waiting for a job.
Now, imagine magnesium as a little dot. We represent the electrons as, you guessed it, dots! So, for magnesium, we'd draw a capital 'Mg' – that's its fancy abbreviation – and then we'd put two dots around it. One on top, one on the bottom, or wherever feels right to you. It's your doodle, after all! These two dots are its valence electrons. Those are the ones that do all the interacting, the ones that get all the attention.
So, there's our lonely magnesium, with its two lonely valence electrons. What are those electrons gonna do? Just float around? Nah, that's boring. Atoms like to be stable, and for most atoms, that means having a full outer shell. It's like wanting to have your full set of Pokémon cards, you know? Complete the collection!

Now, let's meet the other half of our story: oxygen. Oxygen is a bit different. It's a non-metal, and non-metals are often the ones who are looking to grab electrons. They're the collectors, the ones saying, "Ooh, shiny! Gimme!" Oxygen is in Group 16, which means it's got six valence electrons. Six! That's quite a few more than magnesium's two. So, we draw our oxygen atom, 'O', and then we put six dots all around it. You can arrange them however you want, really. Four on the sides, one on top, one on the bottom – whatever makes your artistic soul sing. But those six dots are the key. They're its outer electrons, ready for action.
So, we have magnesium with its two spare electrons, and oxygen with its six, desperately needing two more to feel complete. Do you see where this is going? It's like a perfectly matched pair from a dating app, but way more electrifying. Magnesium is practically begging to get rid of those two electrons, and oxygen is practically singing to take them.
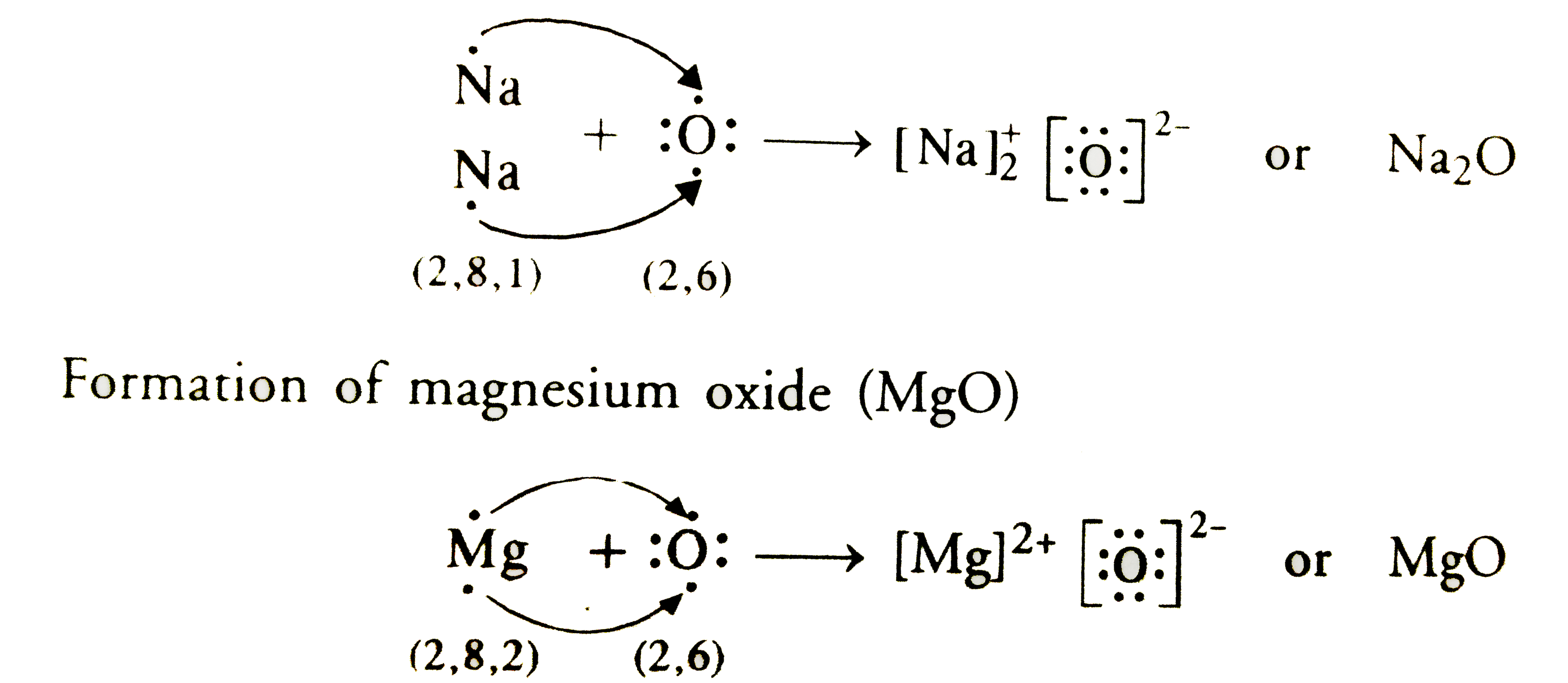
This is where the magic happens. Magnesium, being the generous soul it is, decides to give its two valence electrons away. And who better to give them to than oxygen, who's just dying for a couple of electrons? So, magnesium hands over one electron to oxygen. Poof! Gone. And then, it hands over its second electron. Poof! Also gone.

Now, what happens when an atom loses electrons? It becomes positively charged. Think about it, if you lose something negative, you become more positive, right? So, when magnesium loses those two electrons, it turns into a magnesium ion, and it gets a charge of +2. We write this as Mg2+. It's like it's saying, "I'm all cleaned out! Feel the positivity!"
And what about oxygen? It was happily sitting there with its six electrons, and then BAM! Two more arrive, courtesy of magnesium. Now oxygen has a full outer shell – eight electrons total. It's feeling super stable and happy. But because it gained two negative electrons, it becomes negatively charged. So, oxygen turns into an oxide ion, and it gets a charge of -2. We write this as O2-. It's like, "Yes! I finally have enough! This is the best day ever!"
So, we've got a positively charged magnesium ion (Mg2+) and a negatively charged oxide ion (O2-). What do opposites do? You guessed it: they attract! This attraction is what we call an ionic bond. It's like an invisible magnet pulling them together. They're not actually sharing electrons here, like in some other types of bonds. This is more like a complete transfer. Magnesium said, "Here, take these!" and oxygen said, "Thanks, I'll keep 'em!"

When we draw the electron dot structure for the ionic compound magnesium oxide, it looks a bit different from the individual atoms. We don't really draw dots around the ions anymore in the same way. Instead, we show the ions themselves, with their charges, and we often enclose them in brackets to show they're part of a compound. So, you'd see the Mg2+ ion, and then the O2- ion right next to it. They're stuck together, like glue. Or, you know, like an antacid in your stomach.
Sometimes, people will show the transfer of electrons explicitly in the dot structure before the ions are formed. So, you might see the magnesium with its two dots, and then an arrow pointing those two dots towards the oxygen, which is drawn with its six dots. It's like a little comic strip of the electron transfer. Then, after the transfer, you show the ions. The magnesium ion is just 'Mg2+' inside brackets, and the oxide ion is 'O2-' inside brackets. The dots are now inside the brackets with the oxygen because they belong to the now-stable oxide ion.
It's important to remember that in a solid piece of magnesium oxide, these ions aren't just in pairs. Oh no. They form a giant, repeating structure, like a crystal lattice. Think of it as a really, really big LEGO castle built with alternating positive and negative charges. The Mg2+ ions are surrounded by O2- ions, and the O2- ions are surrounded by Mg2+ ions. It's a beautiful dance of opposite charges, all held together by that strong ionic attraction. It's this strong attraction that makes magnesium oxide a solid at room temperature and gives it a pretty high melting point. It's not gonna melt into a puddle just because you're having a warm day!

So, why do we even bother with these electron dot structures? Well, they're a fantastic visual tool. They help us understand how atoms interact and why they form the compounds they do. It's like learning the alphabet before you can read a book. Once you know the letters (the electrons), you can start to understand the words (the bonds) and then the whole story (the compound).
For magnesium oxide, the electron dot structure clearly shows that magnesium wants to give away two electrons and oxygen wants to gain two. This perfect match is why they form a stable ionic compound. It’s a fundamental concept in chemistry, and honestly, once you visualize it with those little dots, it just clicks.
Think about it: that humble antacid tablet is a testament to the power of electron transfers and ionic bonds! Pretty neat, huh? It’s all about those electrons looking for a stable home. And in the case of magnesium oxide, they found it, in a big, crystal-y way. So next time you pop one of those chalky pills, you can secretly wink at it, knowing the electron dot drama that went into its creation. You're basically a chemistry guru now, my friend. Go forth and explain electron dot structures to anyone who will listen!