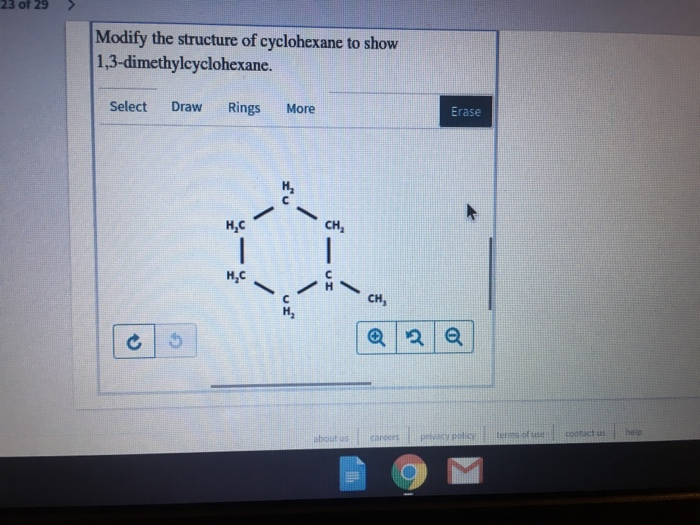
Ever wondered what goes on behind the scenes of the medicines we take, the fuels that power our cars, or even the flavors in our favorite foods? A lot of it comes down to understanding the tiny building blocks of matter: molecules! And today, we're going to have some fun exploring one of them. Learning to draw the structure of 1,3-dimethylcyclohexane might sound a bit technical, but it's actually a fantastic way to get a glimpse into the world of chemistry, and it’s more accessible than you might think!
Why is this particular molecule interesting? Well, cyclohexane rings are like the fundamental skeletons of many important organic compounds. By adding "methyl" groups (which are just carbon atoms with a few hydrogens attached), we create variations that have different properties. For beginners, sketching this molecule is a great first step in visualizing 3D shapes and understanding how atoms connect. It’s like learning to draw simple shapes before you tackle complex art!
For families looking for a hands-on activity, this can be a fun, educational project. Grab some modeling clay or even just paper and colored pencils. It’s a great way to spark curiosity about science. Think of it as a puzzle where you're putting atoms together to form a specific shape. Hobbyists, especially those interested in organic chemistry, molecular modeling, or even creating fictional worlds for stories, will find this a useful skill. Knowing how to represent molecules accurately can add a layer of realism and scientific grounding to your creative endeavors.
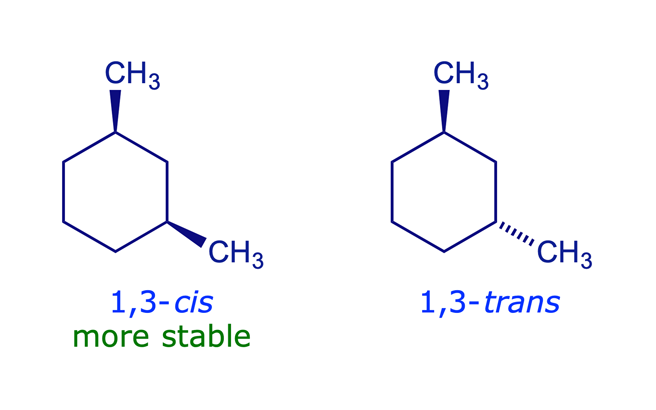
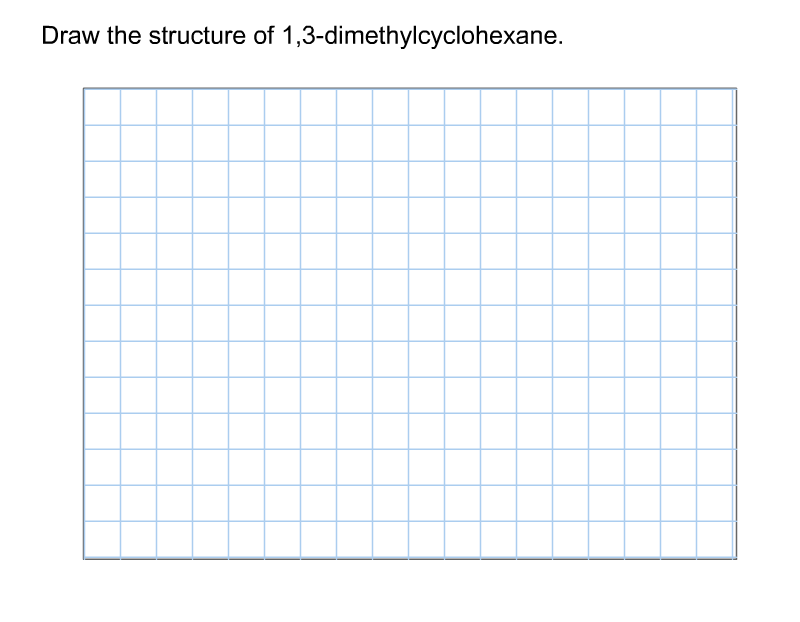
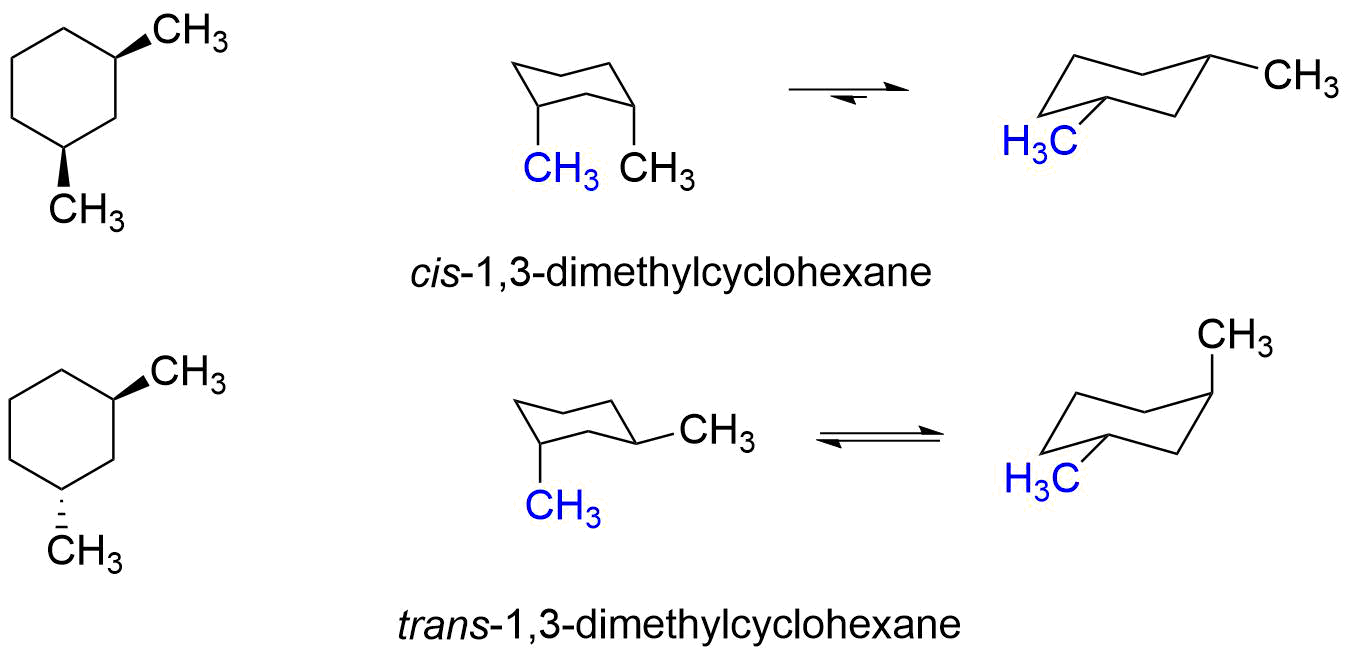
So, what does 1,3-dimethylcyclohexane look like? Imagine a hexagon (that's the "cyclohexane" part), representing six carbon atoms linked in a ring. Now, picture adding two "methyl" groups. The "1,3" tells us where these groups are attached. One methyl group is on carbon atom number 1, and the other is on carbon atom number 3, moving around the ring. Easy peasy, right? There are even variations called stereoisomers, where the methyl groups can point in different directions (up or down relative to the ring), creating molecules that are like mirror images of each other but not quite the same. This adds an extra layer of complexity and is a concept you'll encounter as you delve deeper.
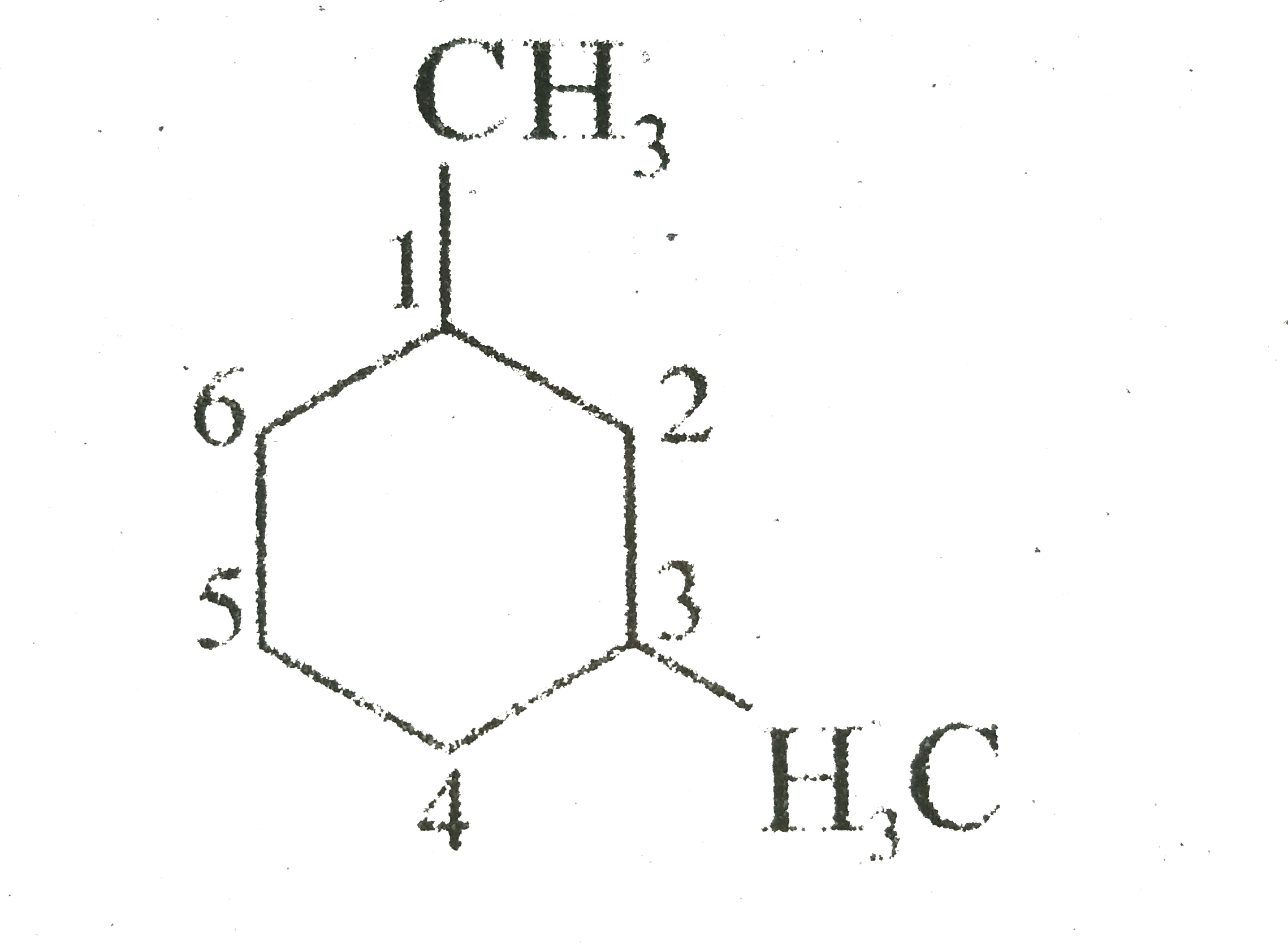
Ready to give it a try? Here are some simple, practical tips:
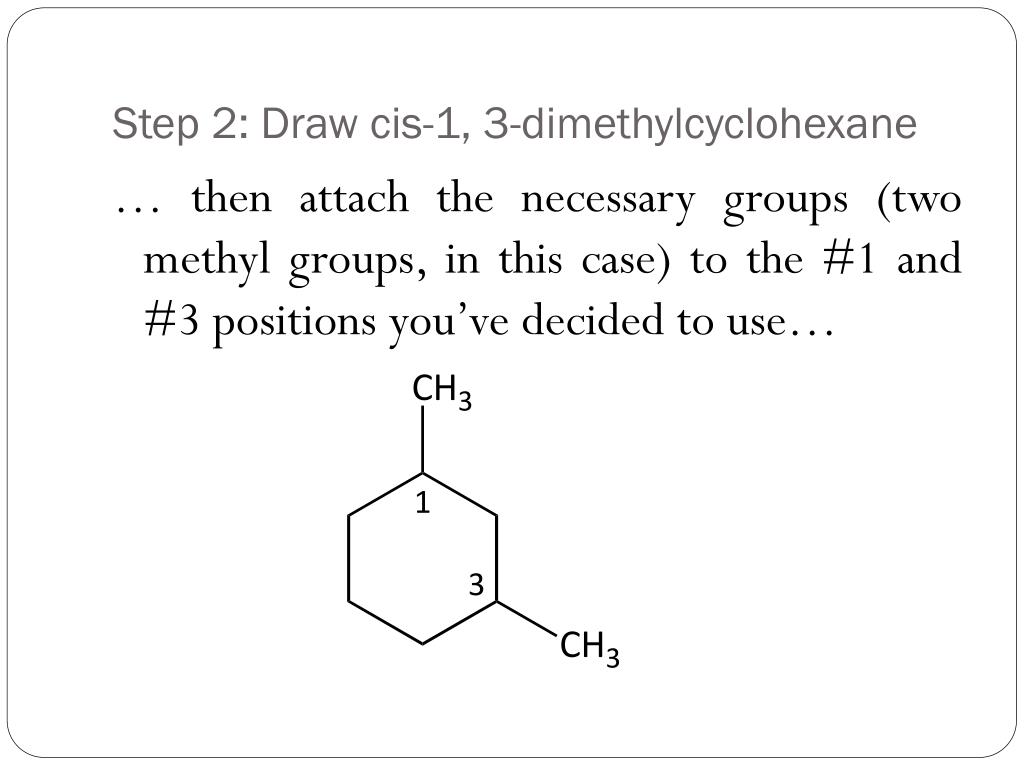
- Start with drawing a hexagon.
- Number the corners of your hexagon from 1 to 6.
- At carbon 1 and carbon 3, draw a small "stick" representing a methyl group (a carbon atom bonded to three hydrogen atoms).
- For a basic representation, you can just draw these bonds pointing outwards.
- If you're feeling adventurous, try drawing the ring in a slightly "wavy" or "chair" shape to represent the actual 3D structure, and then draw your methyl groups pointing up or down.
In conclusion, learning to draw molecules like 1,3-dimethylcyclohexane is a surprisingly enjoyable and valuable skill. It opens up a new way of seeing the world around us, one molecule at a time. It’s a small step that can lead to big discoveries, and most importantly, it’s a lot of fun!