Hey there, fellow science enthusiasts (or maybe just curious cats!)!
So, you've stumbled upon the wonderful world of hydrolysis. Don't let the fancy word scare you off – it's actually pretty straightforward and, dare I say, kinda cool. Think of it like this: hydro means water, and lysis means to break apart. So, hydrolysis is basically using water to break things down. Pretty neat, right? It's like a tiny, molecular demolition crew powered by H2O!
Today, we're going to dive into drawing the products of some hydrolysis reactions. No need to bring out the heavy-duty lab equipment, just your trusty pencil (or stylus, if you're fancy) and a bit of imagination. We're going to keep it light, fun, and hopefully, you'll walk away feeling a little bit smarter and a whole lot more confident about these reactions.
Imagine you've got some molecules chilling out, and then, BAM! Water comes along and says, "Hey, let's shake things up!" Water, bless its simple heart, is made of one oxygen atom and two hydrogen atoms. It's a pretty stable molecule, but it's also got this amazing ability to break the bonds of other molecules. It's like the ultimate party crasher, but in a good way. It breaks things down so they can be rebuilt into something new and useful.
So, the basic idea is that when a molecule undergoes hydrolysis, a water molecule inserts itself into a bond, breaking the original molecule into two smaller pieces. One piece usually picks up a hydrogen atom (H+) from the water, and the other piece picks up the remaining hydroxyl group (OH-) from the water. It's a neat little hand-off of atoms, really. Like passing a baton in a chemical relay race!
Let's start with something super common and relatable: carbohydrates. You know, the stuff that gives us energy – sugars, starches, all that good jazz. When we eat them, our bodies use hydrolysis to break them down into simpler sugars that we can actually absorb and use. Pretty clever, huh? Our bodies are basically tiny hydrolysis factories!
Consider a disaccharide, like sucrose. That's your everyday table sugar. It's made up of two simpler sugars linked together: glucose and fructose. When you add water (via hydrolysis), that bond between glucose and fructose gets broken. So, what do we get? Yep, you guessed it: a molecule of glucose and a molecule of fructose. Easy peasy, lemon squeezy!
Let's visualize this. Imagine sucrose as two building blocks glued together. Water comes along, and with a little nudge, it splits the glue. One building block (glucose) gets a hydrogen atom from the water, and the other building block (fructose) gets the leftover hydroxyl group. And voilà! Two separate, happy building blocks ready to be used.
When you're drawing this, think about the structure of sucrose. It's got a glycosidic bond – that's the fancy name for the link between the two sugar units. In hydrolysis, this glycosidic bond is what gets targeted. Water attacks this bond, breaking it. One sugar gets an -H, and the other gets an -OH. Keep an eye on where those atoms attach; it usually follows some basic bonding rules for carbon and oxygen.
Next up, let's talk about proteins. These are the workhorses of our cells, doing all sorts of jobs. They're made up of smaller units called amino acids, linked together by peptide bonds. When we digest proteins, our bodies break these peptide bonds using hydrolysis. This releases individual amino acids, which our bodies can then use to build new proteins or for other purposes. It's like taking apart a Lego castle to get the individual bricks back!
So, if you have a protein chain, hydrolysis will break the peptide bonds between the amino acids. Each break will add an -H to one amino acid and an -OH to the other. Think of it as splitting an amide bond (which is what a peptide bond essentially is). The nitrogen atom of the original peptide bond will end up with the hydrogen, and the carbonyl carbon will get the hydroxyl group. It’s a classic example of nucleophilic acyl substitution, if you want to get technical, but for drawing purposes, just remember the water molecule splits and adds its parts to the broken bond.
Imagine a long string of beads, each bead representing an amino acid. The string connecting them is the peptide bond. Water comes in, and at each connection point, it snips the string and adds a bit of itself to the ends of the broken beads. So, you end up with individual beads, each now a little different because they've accepted parts of the water molecule.
Drawing the specific amino acids after hydrolysis is where it gets a tiny bit more involved, as each amino acid has a unique "side chain." But the core reaction at the peptide bond is the same: a hydrogen goes to the nitrogen and a hydroxyl goes to the carbonyl carbon. You'll be drawing the structures of the individual amino acids, each with their characteristic R-groups, now liberated from the chain.
Now, let's move on to lipids, or fats. Fats are typically made up of a glycerol molecule and three fatty acid molecules, linked by ester bonds. When fats undergo hydrolysis (like when your body digests them with the help of bile and enzymes), these ester bonds are broken. What do you get? Glycerol and the individual fatty acids. This is crucial because it’s how we get the building blocks for cell membranes and energy storage.
Think of a triglyceride molecule as a three-pronged fork (glycerol) with three long chains (fatty acids) attached to the prongs. Hydrolysis uses water to cut those chains off. For each ester bond that's broken, a water molecule splits. The oxygen atom in the ester bond that was connected to the fatty acid chain will pick up a hydrogen from the water, while the carbonyl carbon of the fatty acid will pick up the hydroxyl group. The glycerol end will also receive a hydrogen and a hydroxyl, essentially regenerating the alcohol functional groups.
So, the products of lipid hydrolysis are glycerol and fatty acids. Glycerol is a simple alcohol with three hydroxyl groups. Fatty acids are carboxylic acids with long hydrocarbon chains. When you're drawing, you'll draw the structure of glycerol and then the structures of however many different fatty acids were attached to the original triglyceride. Remember, each ester linkage gives way to a carboxylic acid group on the fatty acid and a hydroxyl group on the glycerol side.
It’s like a little hydrolysis party for fats! Glycerol is the host, and the fatty acids are the guests. Water comes in, breaks the bonds, and now everyone can mingle separately. The glycerol gets its hydroxyl groups back, and the fatty acids get their carboxylic acid heads.
Let's not forget about nucleic acids, like DNA and RNA. These are super important for life! They're made up of repeating units called nucleotides. Each nucleotide is made of a sugar, a phosphate group, and a nitrogenous base. Hydrolysis can break down nucleic acids into these constituent parts. This is how our cells recycle old RNA or break down DNA.
In nucleic acids, the bonds being broken are phosphodiester bonds and glycosidic bonds. When you hydrolyze a nucleic acid, you can break it down step-by-step. Complete hydrolysis will yield the individual components: a sugar (ribose for RNA, deoxyribose for DNA), a phosphate group, and the nitrogenous bases (adenine, guanine, cytosine, thymine in DNA; adenine, guanine, cytosine, uracil in RNA).
If you're asked to draw the products of partial hydrolysis, you might get shorter strands of nucleotides. But for full hydrolysis, you're looking at those fundamental building blocks. Imagine a DNA double helix. Hydrolysis can snip the phosphodiester backbone, releasing individual nucleotides. Or, it can break the glycosidic bonds, separating the sugar-base pairs from the phosphate-sugar backbone. And then, you can even break down the nucleotides themselves further! It’s like a Russian nesting doll of molecular breakdown.
For drawing, focus on the structures of deoxyribose or ribose (they look similar, but deoxyribose is missing an oxygen on the 2' carbon). Then, you'll draw the phosphate group (PO4^3-). And finally, the nitrogenous bases – these have ring structures and are often the most visually distinct parts of a nucleotide. Each type of base (A, T, C, G, U) has its own unique shape.
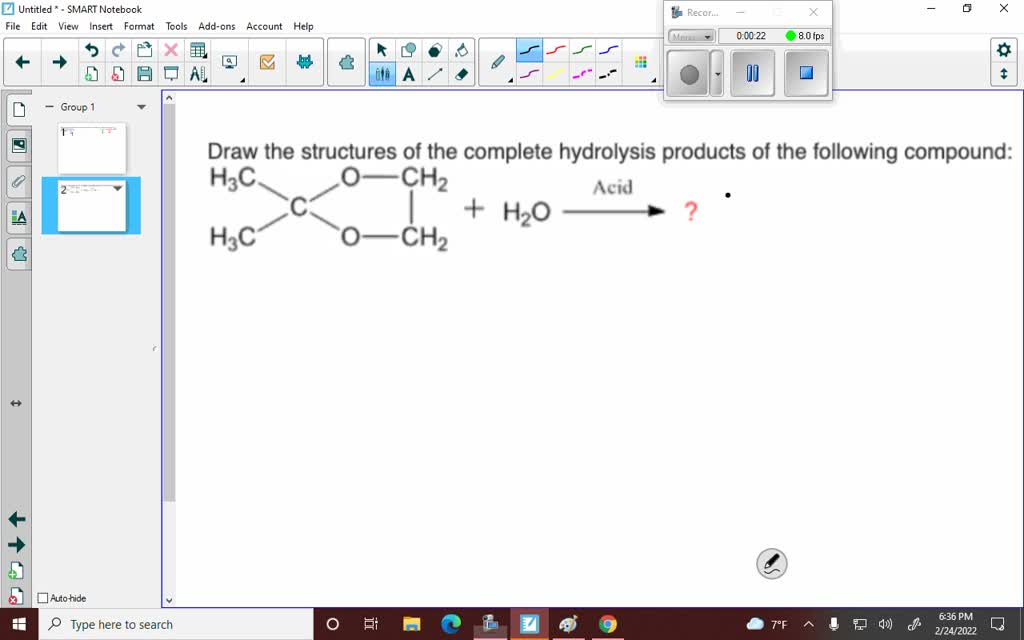
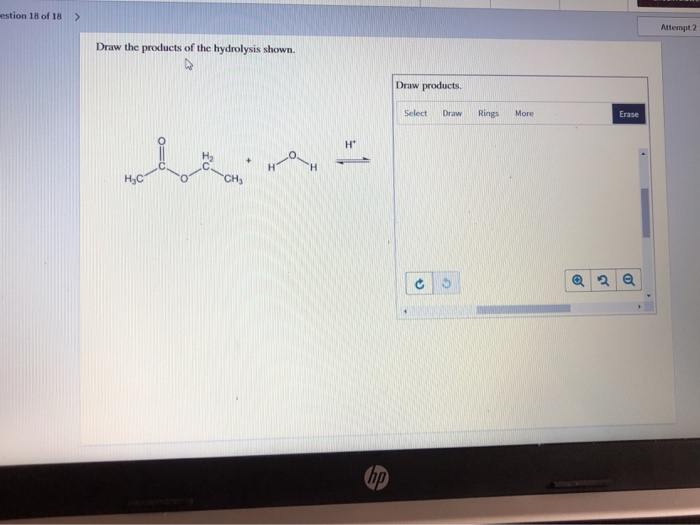
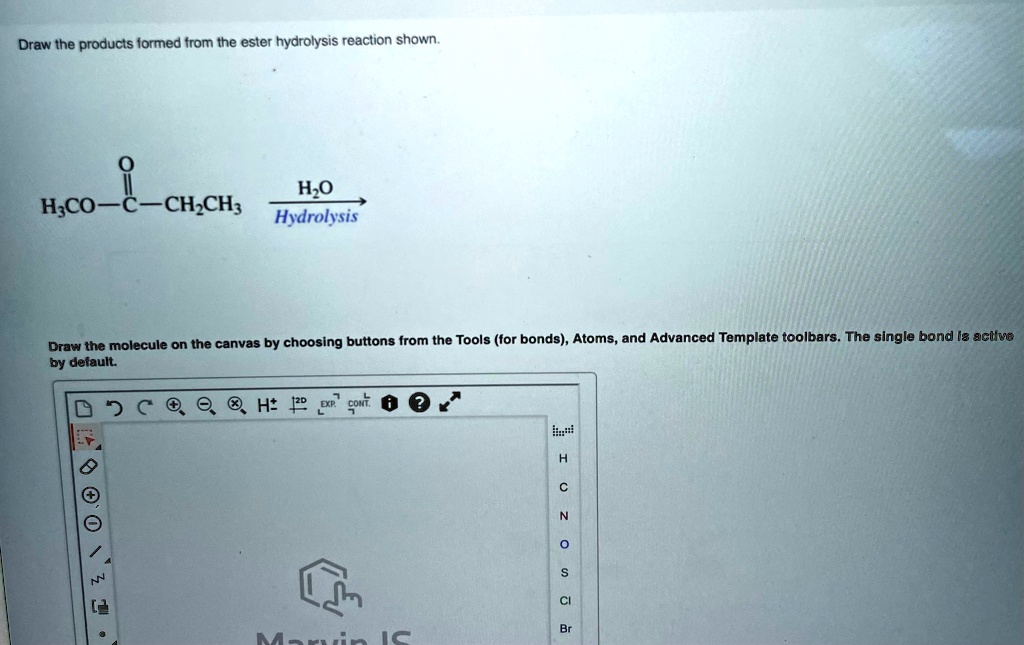
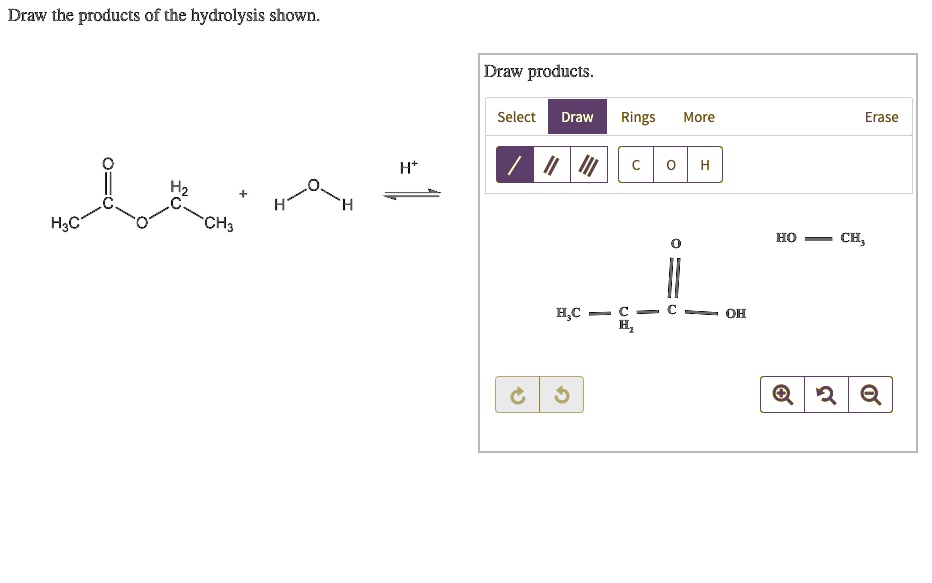
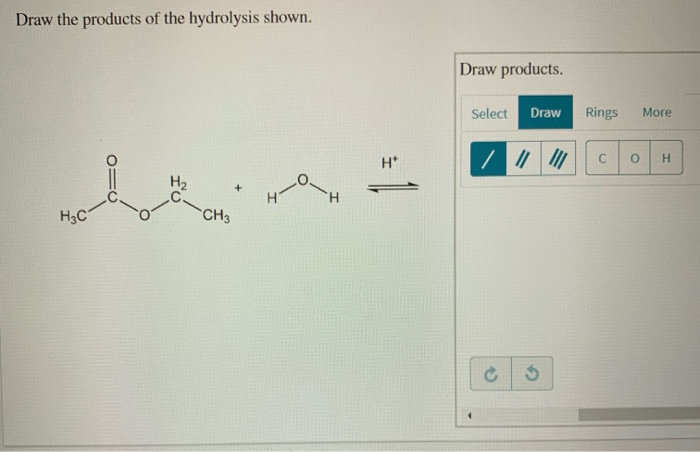
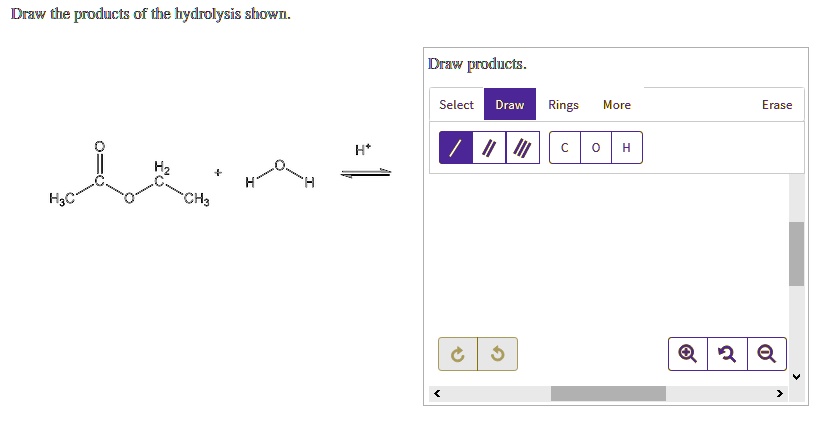
A common hydrolysis reaction you might encounter is the hydrolysis of an ester. We saw this with fats, but esters are found in lots of places. The general rule is that an ester reacts with water to form a carboxylic acid and an alcohol. The bond that breaks is the ester linkage (C-O-C=O). Water inserts itself: the hydrogen goes to the oxygen attached to the alkyl group (forming the alcohol), and the hydroxyl group goes to the carbonyl carbon (forming the carboxylic acid).
Think of it like this: an ester is like a marriage between a carboxylic acid and an alcohol. Hydrolysis is like a divorce, facilitated by water. The two partners are separated, and each picks up a piece of the water molecule to become their own independent entity again. So, the acid part becomes a carboxylic acid, and the alcohol part becomes an alcohol. Simple, right?
For drawing, you'll identify the ester linkage in the starting molecule. Then, you'll draw two new molecules: one with the carbonyl group (C=O) and an -OH attached (that's your carboxylic acid), and the other with the oxygen atom and an -H attached (that's your alcohol). It's a very predictable reaction.
Another interesting one is the hydrolysis of an amide. We touched on this with proteins (peptide bonds are amide bonds). But amides are also found in other synthetic materials, like Nylon. The hydrolysis of an amide also uses water to break the bond, forming a carboxylic acid and an amine. The bond broken is the amide linkage (-C(=O)-N-). Water splits, and the -OH attaches to the carbonyl carbon, while the -H attaches to the nitrogen atom.
So, just like with esters, amides undergo a similar "divorce" with the help of water. The carboxylic acid part and the amine part are set free. If you're dealing with a cyclic amide (like a lactam), hydrolysis will open up the ring to form a molecule that has both a carboxylic acid group and an amine group on the same molecule, separated by the original atoms in the ring.
When drawing, look for the amide bond (-C(=O)-N-). You'll cleave this bond. The carbonyl carbon gets an -OH, and the nitrogen gets an -H. You end up with a carboxylic acid and an amine. Again, it's about identifying the functional group and remembering where the water molecule's atoms go.
Sometimes, you might encounter more complex molecules where multiple hydrolysis reactions can occur. The key is to break down the molecule mentally, identify the bonds that are susceptible to hydrolysis (like ester, amide, glycosidic, phosphodiester bonds), and then apply the simple rule: water comes in, breaks the bond, and adds an -H to one fragment and an -OH to the other.
Don't be afraid to draw it out! Sketching the molecules helps you visualize the process. Label the atoms if it helps you keep track. Think about what kind of functional groups are being formed. Is it a carboxylic acid? An alcohol? An amine? A sugar? Knowing the basic structures of these common organic molecules is a big help.
And remember, even if you're not a chemistry whiz, the underlying principle is about breaking things down with water. It's a fundamental process in chemistry and biology, and understanding it is like unlocking a secret code to how many reactions work. It’s what allows us to digest our food, build new materials, and even understand the building blocks of life itself.
So, the next time you see a hydrolysis reaction, don't panic! Just grab your drawing tools, think about water playing a starring role, and break those bonds. You’ve got this! You’re not just drawing molecules; you’re illustrating the elegant simplicity of how the world around us can be transformed, one water molecule at a time. Keep practicing, and you’ll be a hydrolysis drawing pro in no time. Happy drawing, and remember, even the most complex molecules can be understood by looking at their simpler components!