Hey there! So, you wanna chat about chemistry, huh? Specifically, we're diving into something called the "hydration of 2-butene." Sounds fancy, right? But honestly, it's not as intimidating as it might seem. Think of it like adding water to something, but with a chemical twist. We're talking about 2-butene, which is this super simple molecule, a hydrocarbon. It's got four carbons all lined up, with a double bond in the middle, right between the second and third carbons. Hence, 2-butene. Clever, I know.
And then we're going to "hydrate" it. What does that even mean? Well, in chemistry land, hydration just means adding water. Like when you're feeling parched and you chug a giant glass of water? That's hydration for you. For molecules, it's a bit more precise. We're essentially shoving a water molecule, H₂O, onto that 2-butene. Easy peasy, lemon squeezy, right?
So, why are we even bothering with this? Well, sometimes, in the wild world of organic chemistry, you want to change one type of molecule into another. And adding water to alkenes (that's what things with double bonds are called, by the way – alkenes!) is a classic way to make alcohols. Alcohols are super useful! Think of ethanol, the stuff in your favorite beverage. Or rubbing alcohol. Yep, all alcohols. So, this whole hydration thing is a gateway to making some pretty important stuff.
Now, 2-butene itself, it's got a couple of flavors. You can have cis-2-butene, where the two important side chains (let's call them hydrogens for simplicity here, though they can be other things) are on the same side of that double bond. Like they're chilling together. And then you have trans-2-butene, where they're on opposite sides. Like they're having a little standoff. It’s a subtle difference, but sometimes in chemistry, those subtle differences are a big deal! For this hydration reaction, though, it generally doesn't matter too much which flavor of 2-butene you start with, but it's good to know they exist.
Okay, so we've got our 2-butene, all ready for its water shower. What happens when H₂O comes knocking? Well, water, H₂O, is made of two hydrogen atoms and one oxygen atom. The oxygen is a bit of a diva, you see. It likes to hog the electrons. This makes the O-H bond polar. One side is a little bit negative (the oxygen end), and the other side is a little bit positive (the hydrogen ends). This polarity is key, folks. It’s what makes water such a universal solvent and also what lets it participate in reactions like this one.
Now, the double bond in 2-butene. It's a double bond because there are two pairs of electrons shared between those two carbon atoms. This makes the double bond electron-rich, like a magnet for things that want electrons. Things that are electron-poor, in chemistry terms. And guess what? Those slightly positive hydrogen atoms from the water molecule are just the ticket!
So, in this hydration reaction, we usually need a little help from an acid catalyst. Think of a catalyst as a helpful friend who speeds things up without actually getting used up in the process. They just nudge things along. Acids are good at this because they can provide a source of protons. And what's a proton? It's basically a positively charged hydrogen ion, H⁺. Super electron-poor and ready to party!

The first step in this whole shebang is that the proton from the acid (let's just say it's H⁺ for now) is attracted to that electron-rich double bond of the 2-butene. It’s like the proton sees the double bond and goes, "Ooh, you look like you have snacks!" It attacks the double bond, and a bond forms between the proton and one of the carbons in the double bond. This is a crucial first step.
When this happens, the double bond breaks. Poof! It's now a single bond. But wait, what about that carbon that didn't get the proton? It's now lost an electron connection, and it ends up with a positive charge. We call this a carbocation. It's like a carbon atom that's feeling a little bit lost and a lot positive. It’s an unstable intermediate, meaning it doesn’t hang around for long.
Now, remember our water molecule, H₂O? The oxygen end was a bit negative, right? Well, that negative oxygen is now super attracted to that positively charged carbocation. It’s like the carbocation is crying out for help, and the water molecule is swooping in like a superhero. The oxygen atom of the water molecule, with its lone pairs of electrons, attacks the positive carbon. And bam! A new bond forms between the oxygen of the water molecule and the carbocation.
At this point, we've got our original 2-butene molecule, but now it's got an alcohol group attached. Sort of. The oxygen from the water is bonded to one of the carbons, and the hydrogens from that water molecule are still there. So, it's looking a bit like R-OH, where R is our hydrocarbon chain. We've basically added an -OH group, which is the hallmark of an alcohol, to our molecule.
But there's one tiny little detail. That oxygen atom, in its haste to bond with the carbocation, is now carrying a positive charge itself. It's bonded to two hydrogens and that carbon. So, it's technically an oxonium ion. It’s a bit like saying, "I did it! But now I have a little bit of a glow-up and it's… a bit much."
The final step is where our little proton friend comes back to help again. Or rather, another molecule (could be water, could be the conjugate base of the acid catalyst) comes along and snatches one of those extra hydrogens from the oxonium ion. It's like, "Here, let me take that off your hands!" And with that hydrogen hopping off, the oxygen is finally neutral again, and we have our beautiful, finished alcohol molecule.
So, what do we actually draw? That's the question, right? When we hydrate 2-butene, we can add that -OH group to either of the carbons that were part of the original double bond. Because 2-butene is symmetrical in a way, the double bond is between the second and third carbons. This means we can add the -OH group to the second carbon, or we can add it to the third carbon.
Let's say we add the proton (H⁺) to the second carbon of 2-butene. This means the third carbon ends up with the positive charge. Then, when the water molecule comes in, the oxygen will attach to that third carbon. This gives us a molecule where the -OH group is on the third carbon. The original structure was CH₃-CH=CH-CH₃. If the H⁺ adds to the second carbon, we get CH₃-CH₂-C⁺H-CH₃. Then the water attaches to the C⁺H, giving CH₃-CH₂-CH(OH₂⁺)-CH₃. After deprotonation, we get butan-2-ol! Yep, it’s an alcohol with the -OH group on the second carbon (which was the third carbon in the original numbering if the double bond was between 2 and 3). Wait, my numbering got a little twisted there. Let's clarify!
Okay, let's redraw. 2-butene is CH₃-CH=CH-CH₃. The double bond is between C2 and C3. When the H⁺ adds, it can add to C2 or C3. Let's say it adds to C2. Then C2 becomes CH₃-CH₂⁺. The double bond breaks, and C3 becomes CH₃-C⁺H-CH₃. This is a secondary carbocation, which is pretty stable. Now, if the H⁺ adds to C3, then C3 becomes CH₃-CH₂⁺. The double bond breaks, and C2 becomes CH₃-C⁺H-CH₃. Wait, this is the same carbocation! My bad. This is because 2-butene is symmetrical. The two carbons of the double bond are chemically equivalent in this scenario.
So, when the H⁺ adds, it can add to either carbon of the double bond, and in both cases, you end up with a secondary carbocation, CH₃-C⁺H-CH₃, with the other carbon becoming CH₃-CH₂. So you have CH₃-CH₂-C⁺H-CH₃. This intermediate is the same regardless of which carbon the proton initially attached to, because the molecule is symmetrical. This is why sometimes it’s easier to think about the product directly.
When the water molecule (H₂O) attacks this carbocation (CH₃-CH₂-C⁺H-CH₃), the oxygen will attach to the positive carbon. This gives us CH₃-CH₂-CH(OH₂⁺)-CH₃. After losing a proton, we get butan-2-ol. This molecule has the -OH group attached to the second carbon of a four-carbon chain. It's like, "Hey, I'm butane, but with an alcohol group on the second floor!"
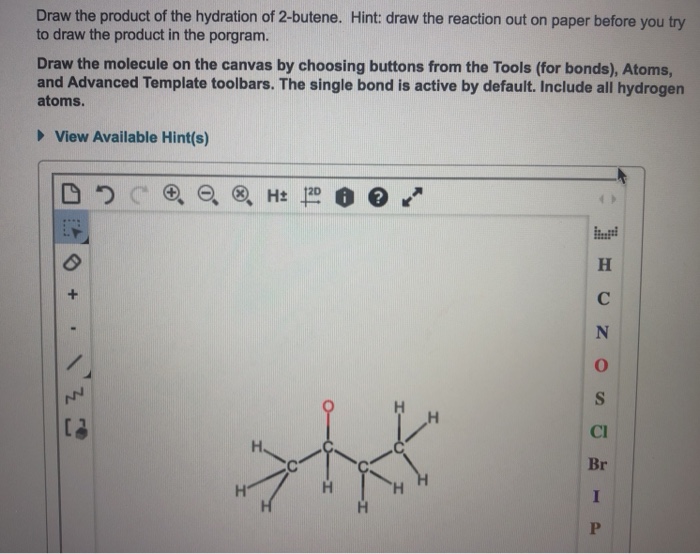
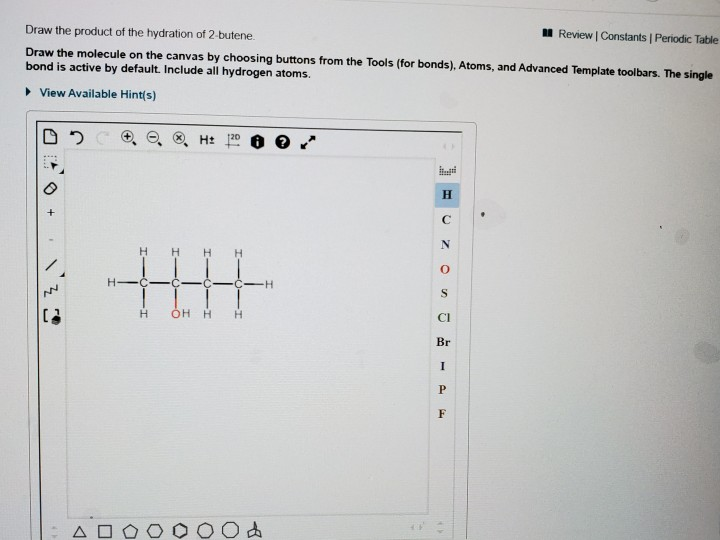
So, when you draw the product, you're drawing butan-2-ol. It's a simple, four-carbon chain. Then, on the second carbon of that chain, you draw an -OH group. You’ll have two CH₃ groups at the ends, and then a CH and a CH₂ in the middle, with the -OH attached to the CH. So, it looks like CH₃-CH(OH)-CH₂-CH₃. See? Four carbons in a row, and the hydroxyl group is hanging out on the second one. It's a perfectly respectable secondary alcohol. No fuss, no drama, just a straightforward chemical transformation.

Now, what if we started with 1-butene? That’s CH₂=CH-CH₂-CH₃. If we hydrate that, things get a little more interesting. The double bond is at the end. The H⁺ can add to the first carbon, making it CH₃-C⁺H-CH₂-CH₃ (a secondary carbocation). Or it could add to the second carbon, making it C⁺H₂-CH₂-CH₂-CH₃ (a primary carbocation, which is less stable). So, nature tends to favor the more stable secondary carbocation. Then, water attacks that, and after deprotonation, you get butan-2-ol again! Or, if the H⁺ adds to the second carbon, and water attacks the C⁺H₂, you'd get CH₃-CH₂-CH₂-CH₂OH, which is butan-1-ol. So with 1-butene, you can get a mixture of butan-1-ol and butan-2-ol. But for 2-butene, because of its symmetry, it's much simpler, and you predominantly get butan-2-ol. It’s like the universe saying, "Let’s keep it neat and tidy with 2-butene, shall we?"
The "drawing" part is pretty standard organic chemistry notation. You'll see skeletal structures, where the carbons and hydrogens are implied at the vertices and ends of lines, and then the -OH group is explicitly drawn. Or you might see expanded structures where all the atoms and bonds are shown. Either way, the key is to show the four-carbon chain and that hydroxyl group sticking out from the second carbon. It's not some mind-bending 3D structure (though you could draw it that way if you wanted to be fancy and show stereochemistry, but that's a whole other coffee chat!). For the basic hydration, just showing the connectivity is usually enough.
So, to recap the drawing. Imagine a line with four segments, representing the four carbons of butane. Then, on the second segment from either end, draw a little hook with an 'O' and an 'H' attached. That's your butan-2-ol. It’s the product of hydrating 2-butene. Simple, elegant, and chemically sound. And the best part? You can draw it without needing a full lab setup, just a pen and some paper!
It’s a testament to how even seemingly complex reactions can be broken down into manageable steps. You have your starting material, your reagent (water, with a little acidic encouragement), and then your product. And in this case, the product is a nice, stable alcohol. No weird byproducts, no explosions (hopefully!), just a smooth transition. Chemistry can be pretty darn predictable sometimes, and that’s a good thing when you’re trying to make things!
So next time you hear "hydration of 2-butene," you can nod knowingly and picture that water molecule making a friendly visit, facilitated by a proton, to create some lovely butan-2-ol. It’s like a molecular spa day, but with chemical reactions. And the drawing? Just a simple chain with a hydroxyl group taking pride of place. Easy as pie. Or, you know, as easy as adding water to an alkene. And that's saying something!