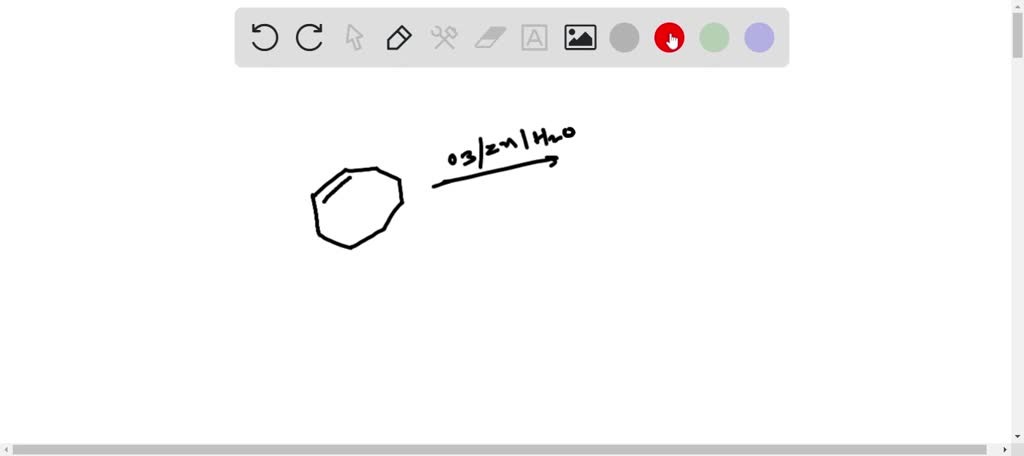
Hey there, chemistry curious folks! Ever wonder what happens when you take a cool, ring-shaped molecule like cyclooctene and give it a little molecular makeover? Well, get ready for some fun because we're about to dive into a reaction that’s like a party for molecules, specifically one called ozonolysis! It’s a fancy name, but trust me, the results are super cool and surprisingly easy to picture.
Imagine cyclooctene is like a perfectly round inner tube, or maybe a giant, squishy, eight-wheeled bicycle wheel. It’s a closed loop, all connected and happy. Now, what if we decided to take scissors and snip that loop right in half? That's kind of what ozonolysis does, but with a super-powered, molecule-eating gas called ozone!
Ozone, you know, the stuff that protects us from the sun's harsh rays way up high in the atmosphere? Well, it’s also a fantastic tool for breaking open those delightful ring structures in organic chemistry. Think of ozone as a tiny, but mighty, molecular demolition crew. They’ve got their sights set on those double bonds, those places where the atoms are holding hands a little too tightly in our cyclooctene friend.
When ozone attacks cyclooctene, it's like a polite but firm intervention. It doesn't go around smashing everything; it targets those specific, juicy double bonds. In cyclooctene, there's one of these special, energetic spots that makes it a bit… bendy and reactive.
So, the ozone molecule, with its three oxygen atoms, swoops in. It's all about breaking that double bond. It’s a bit like the ozone is saying, “Hey, let’s loosen up this connection!” And poof! The double bond snaps, and the ring is no longer a perfect, unbroken circle.
Now, here’s where it gets really exciting. After the ozone does its initial work, it leaves behind some interesting new pieces. These pieces are eager to connect with something else. Usually, in a lab setting, we add a little something to tidy things up and make the final products stable and happy.
For our cyclooctene example, when that eight-membered ring is broken right in the middle where the double bond used to be, it’s like taking that inner tube and cutting it into two equal pieces. Each piece is still an open tube, but now it’s got two ends instead of being a closed loop.
Let’s visualize this! If we take our eight-carbon ring, and imagine the double bond is between carbon number 1 and carbon number 2 (it doesn't really matter which ones we pick for a symmetrical ring like this!), the ozone swoops in and breaks that connection. Suddenly, carbon 1 and carbon 2 are no longer linked to each other through the ring.
Instead, they become the new “ends” of our molecule. And because ozone is a bit of a mischievous molecule, it leaves behind a special structure called an ozonide intermediate. It's like a temporary little hug between the ozone and the broken carbons. But don’t worry, this is just a fleeting moment.
The magic truly happens when we introduce a second step, often involving something like zinc or dimethyl sulfide. This is like bringing in the clean-up crew after the demolition. They help to break apart the ozonide and make everything neat and tidy.

So, what do we end up with when we "draw the ozonolysis product of cyclooctene"? We get two identical pieces! Imagine slicing a pizza right through the middle. You get two big, curved slices. Cyclooctene, when ozonolyzed, gives us two identical open-chain molecules.
Each of these new molecules will have the same number of carbon atoms that were on one side of the original double bond, all linked together in a straight line now. Since cyclooctene has eight carbons, and we're breaking that one double bond, we're essentially cutting it into two equal halves. So, each half will have four carbon atoms.
And what are these four-carbon pieces? They are essentially aldehydes. Aldehydes are super common and important organic molecules. They have a special little functional group, a carbon double-bonded to an oxygen and single-bonded to a hydrogen (we call this a -CHO group). It’s like the molecule’s little waving flag!
So, instead of a closed ring of eight carbons, we get two molecules, each with four carbons, and each ending with that aldehyde group. This means we get octanedial. That’s a mouthful, but think of it as a "double aldehyde" that used to be a ring! It’s like taking a hula hoop, cutting it in half, and finding that each half now has a little handle at both ends!

Let’s break down that name, octanedial. "Octane" tells us there are eight carbon atoms in total in the original molecule’s backbone (even though they're now in two separate pieces). "Dial" tells us there are two aldehyde groups. It’s a perfect descriptor, really, even if it sounds a bit technical.
Think of it this way: cyclooctene is like a delicious, circular donut. Ozonolysis is like taking a super-sharp knife and slicing that donut exactly in half. What do you get? Two perfect semi-circles! In the case of cyclooctene, these semi-circles are molecules, and they have these special reactive aldehyde ends.
So, the product is essentially two molecules of butanal, but because they were originally connected, and we're often talking about the overall result from breaking the ring, we can also think of it as a single molecule called octanedial. Imagine two little four-carbon chains, each with a reactive aldehyde group at one end, and they're just chillin' separately now. They’re no longer tied together in that cyclic fashion.
It’s like taking a necklace made of eight beads in a circle and snapping the clasp. You get two open strands, each with four beads. These strands are the molecules we're talking about, and the "ends" are where the functional groups are.
The beauty of this reaction is its predictability. For a symmetrical molecule like cyclooctene, you know exactly what you're going to get: two identical aldehyde fragments. It's like knowing that if you cut a perfectly round cake in half, you'll always get two equal semi-circles. No surprises, just clean, efficient molecular surgery!
So, when you see cyclooctene and the word ozonolysis, picture that eight-carbon ring getting a friendly but firm intervention. The double bond is broken, the ring opens up, and you’re left with two fabulous aldehyde molecules. It’s a fantastic example of how chemistry can transform structures, turning a cool ring into a linear molecule with exciting new ends. Pretty neat, right?
The final product, essentially, is a molecule with 8 carbons in a row, with aldehyde groups at both ends. It’s like taking that inner tube, cutting it, and then somehow adding a little handle to each of the new open ends. It’s a transformation from a loop to a line, with bonus reactivity!
So, if you ever have to draw the ozonolysis product of cyclooctene, just imagine that perfect circle getting sliced right down the middle. You'll end up with two pieces, each looking like a straight chain of four carbons, with those special aldehyde (-CHO) groups at the ends of each piece. And that, my friends, is the fun and (relatively!) easy ozonolysis of cyclooctene!