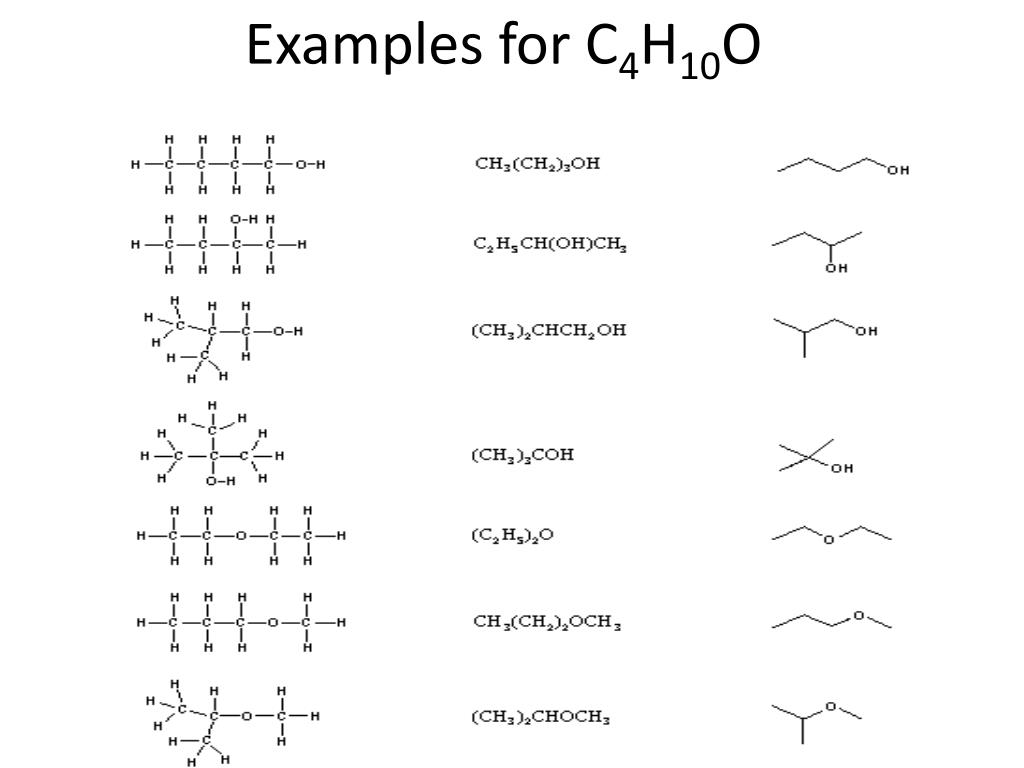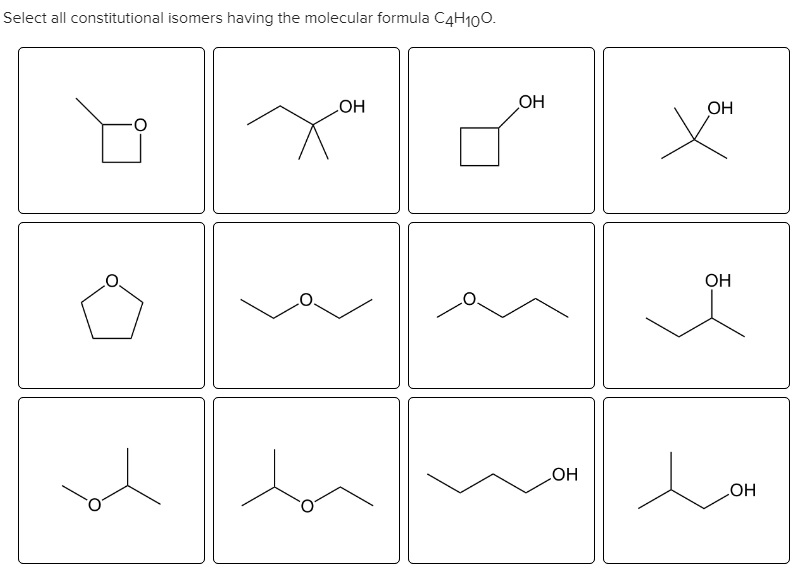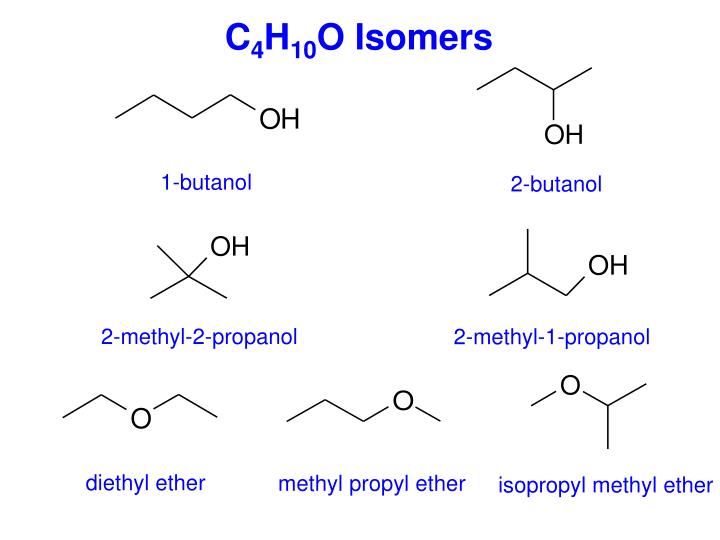
Hey there, fellow curious minds! Ever feel like your brain could use a little… sparkle? A dash of scientific wonder to brighten up your day? Well, get ready, because we're about to dive headfirst into the fascinating, and dare I say, fun, world of molecular doodles! Today, we’re talking about a specific molecular family that’s a real treat to explore: all the ways we can draw out the ethers with the molecular formula C4H10O. Yeah, yeah, I know, "molecular formula" might sound a bit dry, but trust me, it’s like unlocking a secret code to a whole universe of tiny, awesome building blocks.
So, what's the big deal with C4H10O? It’s a humble little combination of carbon (C), hydrogen (H), and oxygen (O) atoms. But here’s the cool part: even with this simple formula, nature (and us chemists, of course!) can arrange these atoms in loads of different ways. It’s like having a set of LEGOs – the same pieces can build a spaceship, a castle, or a really, really happy dog. And when it comes to C4H10O, these different arrangements are called isomers. Think of them as molecular cousins, all related but with their own unique personalities and properties. Pretty neat, right?
Now, before your eyes glaze over, let’s talk about what an "ether" actually is in this context. In chemistry, an ether is a molecule where an oxygen atom is sandwiched between two carbon atoms. Like a tiny molecular hug! So, for our C4H10O friends, we’re looking for structures that have this C-O-C connection, and where the remaining atoms fill up the rest of the space to meet that C4H10O target. Sounds straightforward enough, but the magic happens in where those carbons go!
Let’s grab our metaphorical pencils (or, you know, just use our imaginations!) and start sketching. The simplest way to arrange four carbons and an oxygen in an ether structure is to have a straight chain of carbons. Imagine a little string of four beads. Now, stick an oxygen in there somewhere in the middle, and then add hydrogens to fill everything up.
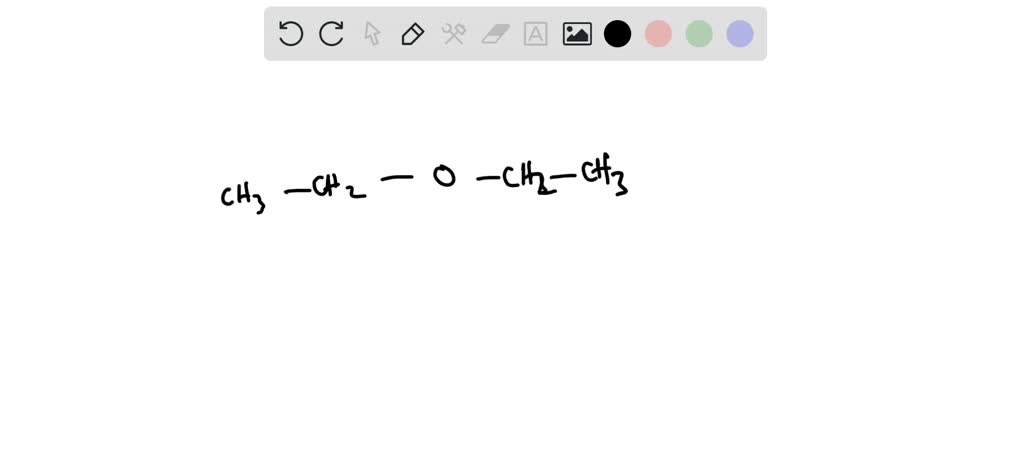
Our first contender, and perhaps the most straightforward, is diethyl ether. This one’s a classic! Think of it as two ethyl groups (that’s two carbons with hydrogens attached) holding hands via an oxygen atom. So, you’ve got CH3-CH2-O-CH2-CH3. See? Two carbons on one side of the oxygen, two on the other. It’s symmetrical, clean, and oh-so-ether-y. Fun fact: diethyl ether used to be a pretty common anesthetic! Imagine, a molecule so simple, yet capable of such a profound effect. It’s like a tiny chemical superhero, in its own quiet way.
But wait, there’s more! We don’t have to keep our carbon chains perfectly straight. We can start branching out. Think of it like a tree – some branches are long and straight, others have little offshoots. This branching leads to a whole new set of possibilities for our C4H10O ethers.

Consider methyl propyl ether. Here, we have a single methyl group (that’s just a CH3) attached to one side of the oxygen, and a propyl group (that’s three carbons) attached to the other. But here’s where it gets interesting: that propyl group can itself be branched! So, we could have a straight propyl chain: CH3-O-CH2-CH2-CH3. Or, we could have a branched propyl chain, leading to methyl isopropyl ether. In this case, the propyl group is like a little 'Y' shape: CH3-O-CH(CH3)2. It’s the same number of atoms, the same ether linkage, but the shape is different. It’s like two people wearing the exact same outfit but styled slightly differently – still recognizable, but with their own flair.
And what about the possibility of the oxygen being at the end of a branch? Well, if the oxygen is at the very end of a chain, it's not an ether anymore! That would make it an alcohol, like butanol or tert-butanol. So, to be an ether, that oxygen must be connected to two carbons. It’s a rule of the molecular club!
Let’s keep exploring those branches. If we have a propyl group on one side, we said it could be straight or branched. What if we have two branches on the other side of the oxygen? This leads us to the wonderfully named di-isopropyl ether. Wait a minute… di-isopropyl? That’s four carbons on both sides of the oxygen, each in an isopropyl arrangement. CH(CH3)2-O-CH(CH3)2. But that only adds up to four carbons and an oxygen! Hmm, did I lose some hydrogens along the way? Ah, yes, the formula is C4H10O. Di-isopropyl ether would actually be C6H14O. My apologies, a momentary brain fog! This is why drawing them out is so important, folks! We're looking for C4H10O. So, no di-isopropyl ether for our current quest. Back to the drawing board… or rather, the molecular structures!
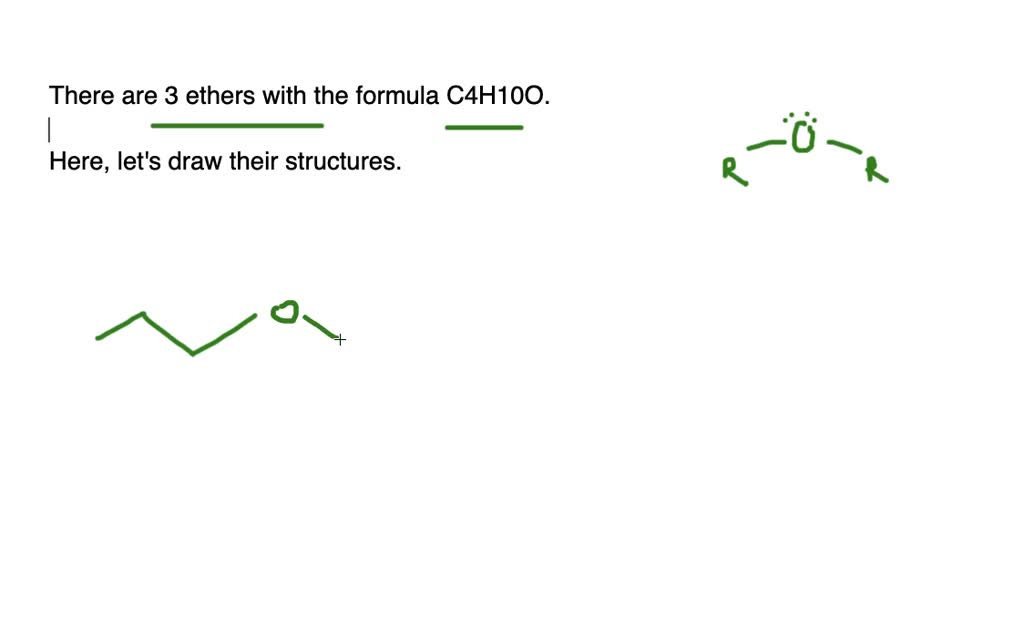
Let’s reconsider our branching. We had diethyl ether (straight chain on both sides). We had methyl propyl ether (methyl on one side, straight propyl on the other). We had methyl isopropyl ether (methyl on one side, branched propyl on the other). Are there any other ways to arrange those four carbons around the oxygen while keeping it an ether and adhering to C4H10O?
Think about the propyl group again. It has three carbons. We can have a straight chain (propane), or we can branch it. If we have a straight chain of three carbons, that's a n-propyl group. If we branch it, so the middle carbon has two other carbons attached, that's an iso-propyl group. So, when we have methyl isopropyl ether, we've used up our branching potential in that propyl group.
What if we consider the placement of the oxygen? In diethyl ether, it’s in the middle of a four-carbon chain. In methyl propyl ether, it’s at the end of a three-carbon chain, next to a one-carbon chain. What if we put the oxygen in a different spot within a branched four-carbon structure?
Let’s go back to the four-carbon skeleton. We can have a straight chain of four carbons. Or, we can have a three-carbon chain with a one-carbon branch. This branched four-carbon skeleton is called isobutane. Now, can we insert an oxygen into this branched skeleton to form an ether?
If we have the isobutane skeleton (a central carbon bonded to three other carbons, with hydrogens filling in), and we want to insert an oxygen, where can it go to make an ether (C-O-C)? We can’t just stick it anywhere without breaking the carbon skeleton. Instead, we think about the groups attached to the oxygen. We have a total of four carbons to distribute on either side of the oxygen.
We've covered the 2-carbon + 2-carbon split (diethyl ether). We've covered the 1-carbon + 3-carbon split (methyl propyl ether and methyl isopropyl ether). Are there any other ways to divide those four carbons into two groups attached to an oxygen?
Let's look at the structures again. Diethyl ether is C2H5-O-C2H5. Methyl propyl ether is CH3-O-C3H7. Methyl isopropyl ether is CH3-O-CH(CH3)2.
It seems like we’ve pretty much exhausted the possibilities for drawing out all the ethers with the molecular formula C4H10O! We have structures that are symmetrical (diethyl ether) and asymmetrical (methyl propyl ethers). We have straight chains and branched chains. It’s a beautiful demonstration of how atoms, with just a few simple rules, can create such diverse molecular personalities.
Why is this fun, you ask? Because it’s like solving a tiny, delightful puzzle! It’s about understanding the underlying logic of the universe at its most fundamental level. And once you start seeing these molecular structures, you start seeing them everywhere. In the fuels that power our cars, in the solvents used in laboratories, and even in the scents of certain fragrances. It’s a secret language of the world around you.
So, the next time you encounter something with a bit of an interesting aroma, or you’re reading about a chemical process, remember these little C4H10O ethers. Remember how their simple building blocks can be arranged in so many clever ways. It’s a reminder that even in the seemingly complex world of science, there’s a profound beauty and elegance to be found. Keep looking, keep questioning, and you might just find a whole new world of fun waiting to be discovered!
Isn’t that inspiring? The universe is literally built from these tiny, incredible structures. And the more you learn about them, the more you realize there is to explore. So, don’t be afraid to dive deeper, to doodle your own molecules, and to let your curiosity guide you. Who knows what amazing discoveries and delightful insights await you on your scientific journey!