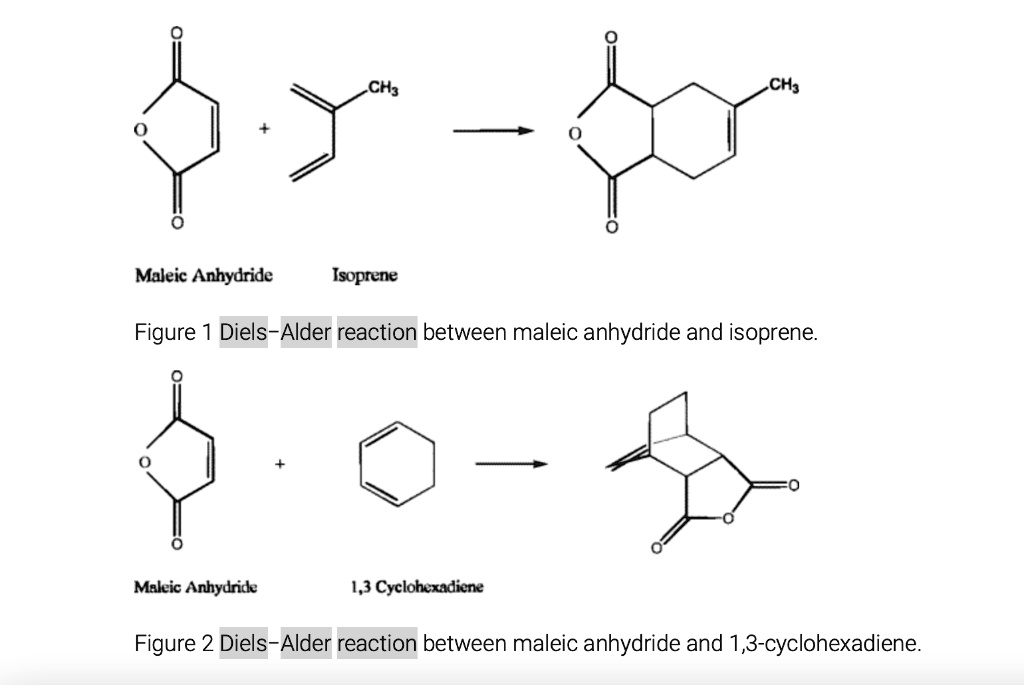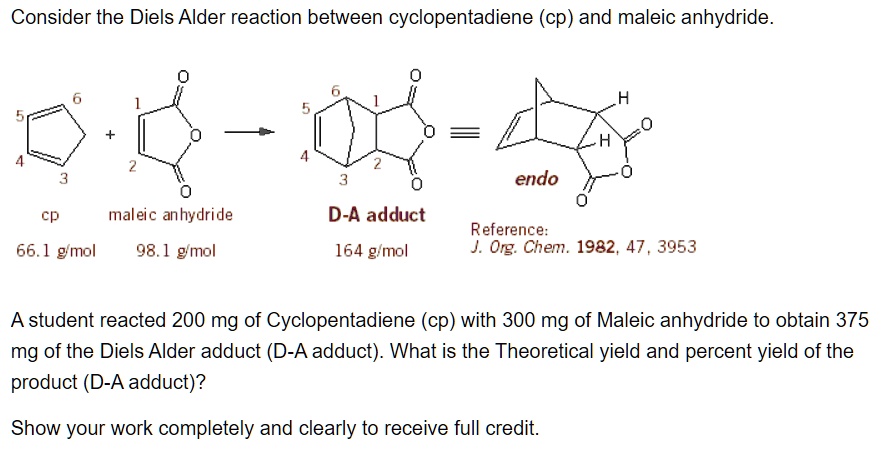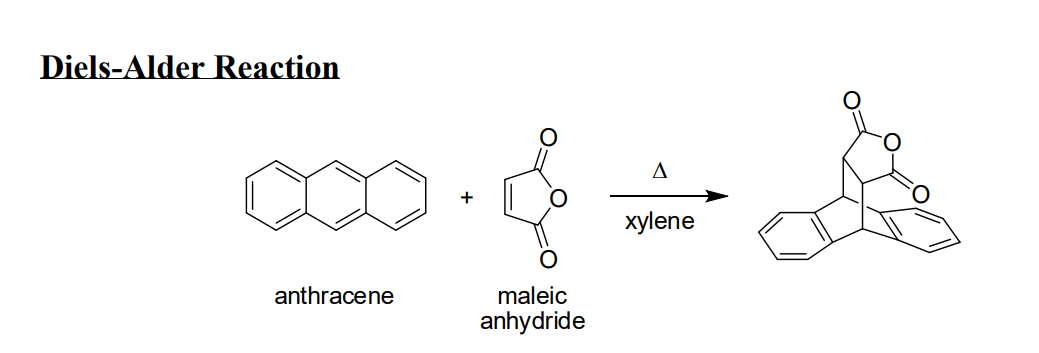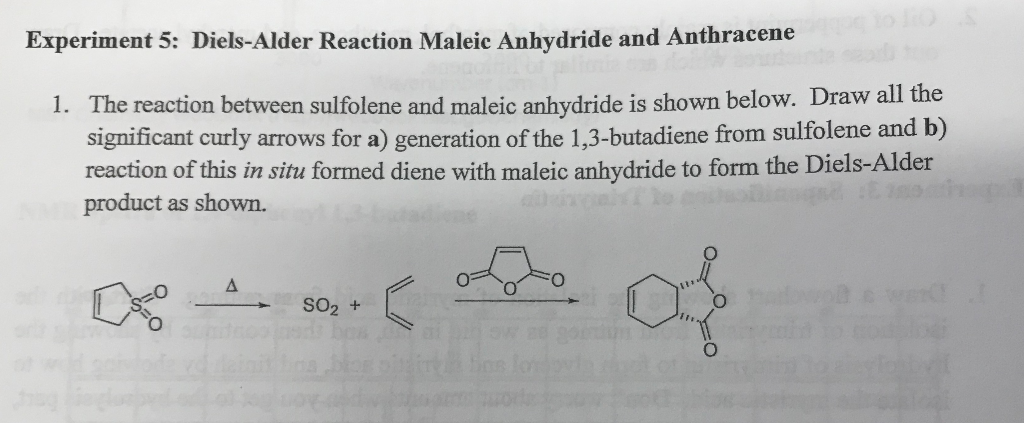
Ever wondered how scientists build complex molecules, the building blocks of everything from your morning coffee to life-saving medicines? Well, get ready to discover a chemical party trick that's as elegant as it is powerful! We're diving into the world of the Diels-Alder reaction, specifically when our star player, maleic anhydride, jumps into the ring. Think of it as a super-efficient way to stitch two molecules together, creating a new ring structure with remarkable ease. This isn't just some obscure lab experiment; it’s a fundamental reaction that underpins the synthesis of countless important compounds. So, buckle up, because we're about to explore why this particular chemical dance is so incredibly fun and useful!
The Magic of Making Rings
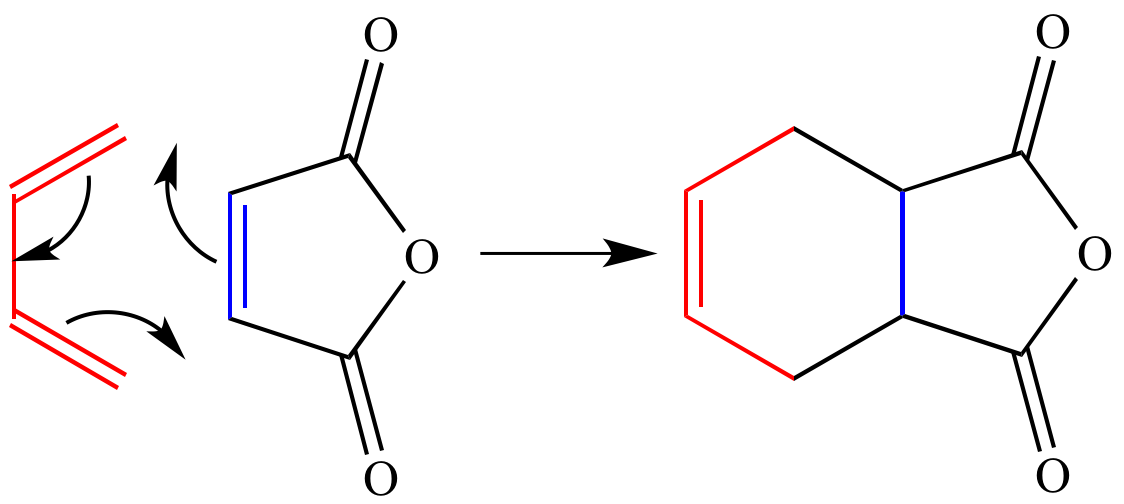
At its heart, the Diels-Alder reaction is a special kind of chemical handshake between two types of molecules: a diene and a dienophile. Imagine a diene as a molecule with two double bonds in a specific arrangement, ready to offer up some electrons. The dienophile, on the other hand, is a molecule with a double or triple bond that’s eager to accept those electrons. When these two meet under the right conditions, they don't just stick together; they elegantly form a brand-new six-membered ring structure. It’s like a perfectly choreographed dance where two partners come together to create something entirely new and stable. This ring-forming ability is what makes the Diels-Alder reaction so incredibly valuable in organic chemistry.
Now, let's talk about our special guest star: maleic anhydride. This molecule is a classic example of a highly reactive dienophile. It’s got a double bond that’s just begging to be involved in this ring-forming reaction. When maleic anhydride pairs up with a suitable diene, it doesn't just form any ring; it forms a specific type of ring that often contains a carboxylic acid or anhydride functionality. This is where the magic really kicks in because these functional groups are incredibly versatile. They can be easily modified further, allowing chemists to build even more complex and diverse molecules. Think of it as getting a fantastic starter kit with a special tool already included, making your molecular construction project much easier.
Why is This Reaction So Swell?
So, why is this particular chemical reaction so popular and useful? There are several fantastic reasons:

- Efficiency: The Diels-Alder reaction is renowned for its atom economy. This means that most of the atoms from the starting materials end up in the final product. Very little is wasted, making it an environmentally friendly and practical choice for synthesis. It’s like a chef who uses every part of an ingredient, leaving no scraps behind!
- Selectivity: This reaction is often highly selective, meaning it tends to form a specific product or a limited set of products. This is crucial in chemistry because we often want to create a very particular molecule without a messy mix of unwanted byproducts. The Diels-Alder reaction’s predictability makes it a reliable tool.
- Stereochemistry Control: Even more impressively, the Diels-Alder reaction can often control the spatial arrangement of atoms in the new ring. This is known as stereochemistry, and it's incredibly important because different spatial arrangements of the same atoms can lead to compounds with drastically different properties. For example, one stereoisomer might be a beneficial drug, while another might be inactive or even harmful. The Diels-Alder reaction provides a way to build these intricate structures with precision.
- Versatility: The sheer variety of dienes and dienophiles that can participate means this reaction can be used to create an astonishing range of cyclic compounds. This versatility makes it a cornerstone for synthesizing everything from natural products and pharmaceuticals to polymers and advanced materials.
When maleic anhydride is the dienophile, it brings its own set of advantages. Its electron-deficient double bond makes it a very reactive partner for many dienes. The anhydride group it introduces is a powerful functional handle. It can be readily hydrolyzed to form two carboxylic acid groups, or it can undergo various nucleophilic attacks, opening up a world of further chemical transformations. This makes it an ideal starting point for building complex carbon skeletons with desired functionalities.
"The Diels-Alder reaction is like a master key that unlocks the door to countless molecular structures."
Imagine you need to create a new type of plastic with specific properties. Or perhaps you're designing a new drug molecule that needs a particular ring structure to interact with a target in the body. The Diels-Alder reaction, especially with a workhorse like maleic anhydride, provides a straightforward and elegant solution. It’s a reaction that scientists have been exploring and utilizing for decades, constantly finding new and innovative ways to apply its power. It's a testament to its fundamental importance and enduring appeal in the world of chemical synthesis. So, the next time you hear about chemists building complex molecules, remember the elegant dance of the Diels-Alder reaction, with maleic anhydride often leading the way, proving that even in chemistry, forming strong bonds can be a truly beautiful and useful endeavor.