Hey there, coffee buddy! So, we're diving into the weird and wonderful world of organic chemistry today, aren't we? Specifically, we're gonna chat about this little guy called the cycloheptatrienyl anion. Sounds fancy, right? Like something you’d find in a sci-fi movie. But trust me, it's a lot less laser guns and a lot more… electrons. And the big question on everyone’s mind, or at least, my mind when I’m procrastinating, is: is this thing aromatic or not? It’s a total cliffhanger, I know!
Now, what even is aromaticity? It’s basically chemistry's way of saying something is super stable, like it’s found its zen. Think of benzene. Everyone knows benzene, right? That cool, six-membered ring with alternating double bonds. Benzene is the poster child for aromaticity. It’s got this delocalized cloud of electrons zipping around, and it just makes everything so happy and stable. No wonder it’s so common!
So, how do we figure out if our cycloheptatrienyl anion, which we’ll call the tropylium anion for short – much easier to say, don't you think? – is in the same chill-out club as benzene? Well, there are a few rules, like a secret handshake for aromaticity. You gotta follow them, or you’re out, buddy.
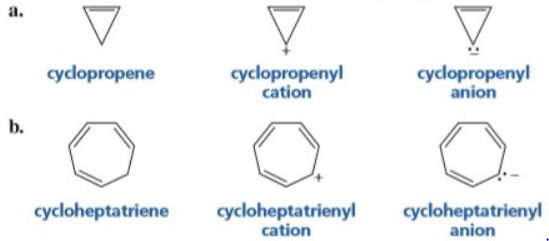
First up, you need a ring. Easy enough. Our tropylium anion has a seven-membered ring. Seven! That’s already a bit of a deviation from the classic six, isn't it? Adds a little spice, maybe? Or is it just… more potential for chaos? We’ll see.
Next, this ring needs to be planar. Like, flat. As flat as your chances of acing that organic chemistry exam without studying. This is crucial because for those electrons to get all delocalized and happy, they need to be able to move freely in a flat plane. If the molecule is all warped and wobbly, the electrons get stuck in their little lanes, and nobody’s happy.
And then, the big one. The Hückel’s Rule. This is where things get numerical, and honestly, sometimes I feel like I need a calculator just to understand chemistry. Hückel’s Rule says you need a specific number of pi electrons. And that number is 4n + 2, where ‘n’ is a nice, whole number. So, if ‘n’ is 0, you get 2 pi electrons. If ‘n’ is 1, you get 6 pi electrons. If ‘n’ is 2, you get 10 pi electrons. See the pattern? It’s always 2, 6, 10, 14… you get the drift. It’s like a secret code for stability.
So, let’s break down our tropylium anion. We have our seven-membered ring. Check. Is it planar? This is where it gets a little murky. For a while, scientists weren’t entirely sure. Some calculations suggested it might not be perfectly flat. Imagine trying to do a synchronized dance on a slightly lumpy trampoline. Not ideal for everyone getting their moves in sync, right? But then, better techniques and more sophisticated thinking came along, and it turns out, yeah, it can be pretty darn planar. Especially when it’s being all… anion-y.
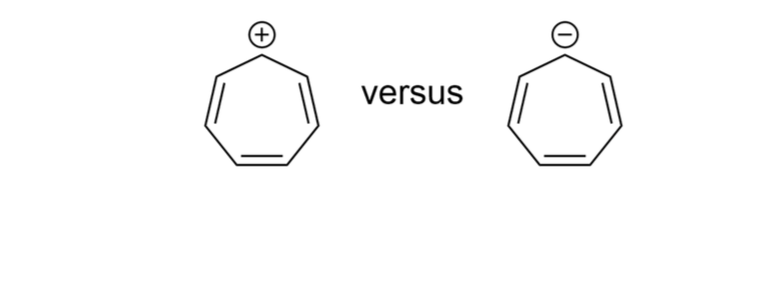
Now for the pi electrons. This is the juicy part. Our tropylium anion has a negative charge. And what does a negative charge often mean in organic chemistry? Extra electrons, my friend! It’s like finding a twenty-dollar bill in an old coat pocket. A welcome surprise. So, the neutral cycloheptatriene molecule has three double bonds. That gives you 3 x 2 = 6 pi electrons. Pretty good start, right? That's the magic number 6 (when n=1 in Hückel’s Rule). So, the neutral cycloheptatriene? It’s pretty stable, actually. It's got some aromatic character, but it's not fully aromatic in the way benzene is.
But we’re talking about the anion here! When it becomes an anion, it gains an extra pair of electrons. Where do they go? They get to join the pi system. So, instead of 6 pi electrons, we now have 6 + 2 = 8 pi electrons. Uh oh. Eight. Does 8 fit into the 4n + 2 club? Let’s see. If 4n + 2 = 8, then 4n = 6, and n = 6/4, which is 1.5. And 1.5? Not a whole number. Nope, sorry, you’re not invited to the party, eight pi electrons. This is where the tropylium anion starts looking a little less aromatic and a lot more… something else.

So, if it’s not following Hückel’s Rule, what is it? Well, molecules that have a number of pi electrons that doesn't fit the 4n + 2 rule, but are still cyclic and have a delocalized pi system, are often called antiaromatic. And antiaromatic? That's the opposite of aromatic. It’s like the molecule is actively trying to be unstable. It’s high-strung, jumpy, and just generally a bit of a mess. Not exactly the chill vibes we were hoping for.
So, the story goes: tropylium anion has 8 pi electrons. It’s cyclic. It’s (mostly) planar. But 8 pi electrons? That’s a big NO for aromaticity. That makes it… you guessed it… antiaromatic. Can you believe it? All that potential for stability, and it ends up being the exact opposite! It’s like you’re baking a cake for a special occasion, and instead of rising beautifully, it just collapses into a sad, flat puddle. Disaster!
However, nature, as always, loves to throw us a curveball. The thing is, true antiaromaticity is usually super unstable. Like, “I’d rather explode than exist” unstable. So, for a molecule to be truly antiaromatic, it has to be forced into it. And even then, it’ll do everything in its power to not be planar. It’ll warp and twist out of shape to break that delocalized pi system, just to avoid being that unstable antiaromatic state. It’s like our tropylium anion is doing a little jig to avoid the dreaded antiaromatic label.
And this is where the story of the tropylium anion gets even more interesting. While the simple counting of pi electrons suggests it’s antiaromatic, the reality is a bit more nuanced. Because seven-membered rings have this flexibility, the tropylium anion can actually adopt a slightly non-planar shape. This distortion breaks the continuous pi system, and guess what? It avoids being fully antiaromatic! It’s like it’s saying, “Oh, you want me to be unstable? Nope, I’m going to be a little bent, thank you very much.”

So, it’s not perfectly planar, and therefore, the pi electrons aren't fully delocalized in the way they need to be for classical antiaromaticity. This means it’s not the super-duper unstable antiaromatic compound that Hückel’s Rule might initially suggest. It’s more like it’s sitting on the fence, or maybe doing a graceful pirouette away from the edge of the antiaromatic cliff.
Some chemists might even argue that because it avoids the truly unstable state by distorting, it’s not really antiaromatic. It’s more like… non-aromatic. Non-aromatic is the ultimate shrug. It's not aromatic, and it's not antiaromatic. It's just… there. Like that sock that goes missing in the laundry. It’s not a disaster, but it’s not particularly noteworthy either. It just is.
The key here is that true antiaromaticity is so energetically unfavorable that molecules will often distort to break the planarity and the continuous pi system. So, while it has 8 pi electrons, the tropylium anion, due to its ring size and flexibility, can avoid the full consequences of Hückel’s Rule for antiaromaticity by becoming non-planar. It’s a clever escape!

Think about it: if it were perfectly planar and antiaromatic, it would be incredibly reactive and probably wouldn’t hang around for long. But we can observe it, and it’s not like it’s spontaneously combusting. So, that tells us something is up. It’s not the epitome of aromatic stability, but it’s also not the poster child for chemical chaos. It’s somewhere in the middle, playing it safe.
So, to sum it all up, the tropylium anion has 8 pi electrons. By the strict letter of Hückel’s Rule, this would point towards antiaromaticity. BUT! And this is a big, fat, chemistry-nerd BUT, real-world molecules are a bit more complicated. The seven-membered ring can pucker, breaking the perfect planarity. This distortion helps it avoid being the hyper-unstable antiaromatic species. So, while it could be antiaromatic, it chooses not to be. It ducks out of the way.
Therefore, is the cycloheptatrienyl anion aromatic? Absolutely not. Is it truly antiaromatic in the sense of being extremely unstable and forced into that state? Most chemists would say no, not really. It leans more towards the non-aromatic category because it avoids the unstable antiaromatic configuration by distorting. It’s a survivor, a molecule that knows when to bend the rules (literally!) to keep itself from falling apart. Pretty neat, huh? Chemistry can be like a thrilling detective novel sometimes, full of twists and turns!
So, next time you’re sipping your coffee and contemplating the mysteries of the universe, you can ponder the plight of the cycloheptatrienyl anion. Is it or isn’t it? The answer, as with so many things in science, is a delightful “it depends,” with a healthy dose of molecular gymnastics thrown in for good measure. Cheers to that!