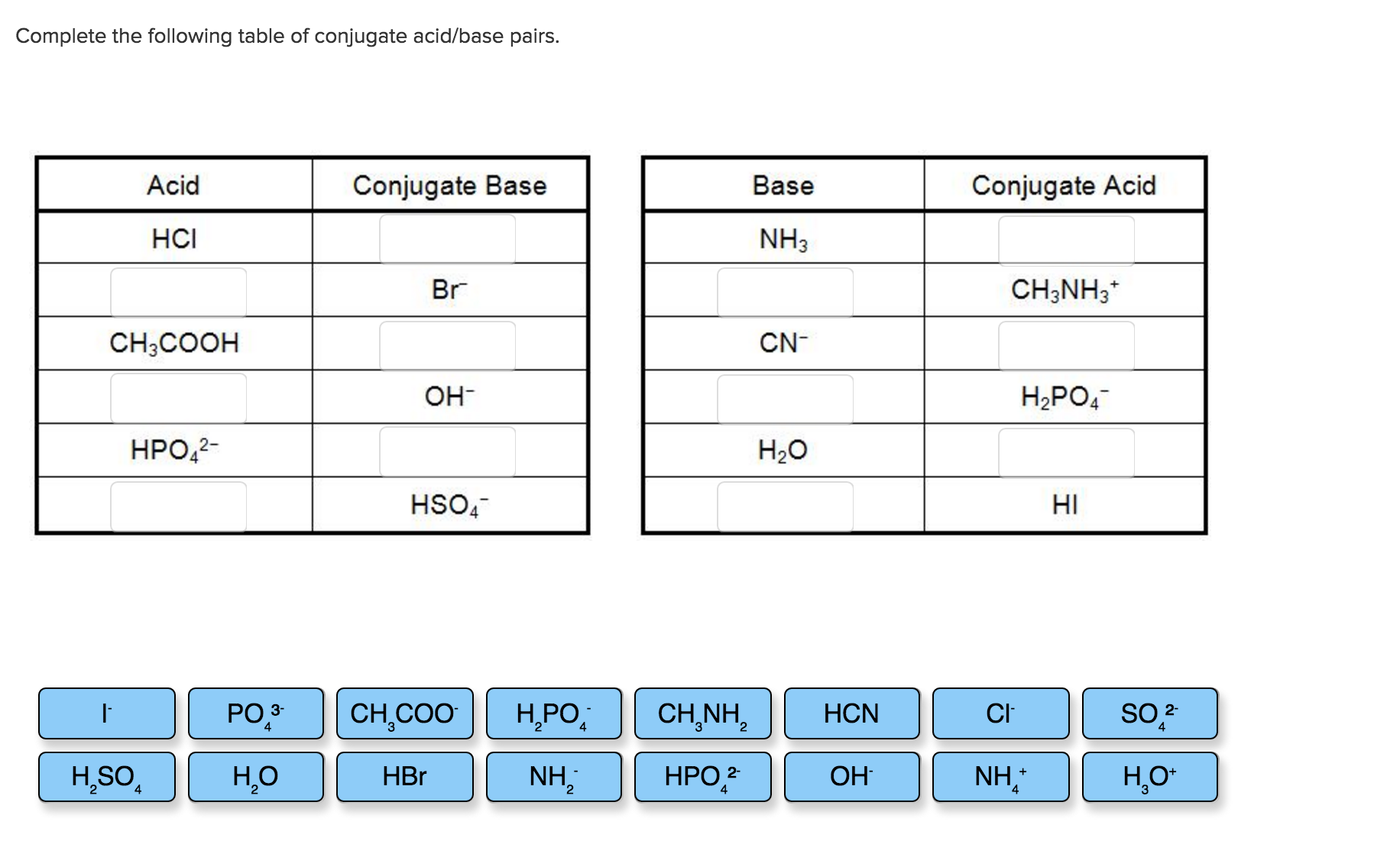
Alright, folks, gather 'round! Let's talk about something that might sound a little… intense. We're diving into the wild world of conjugate acid-base pairs. Think of it like a chemistry matchmaking service, but way less awkward and with more potential for explosions. Okay, maybe not explosions. But definitely some interesting reactions!
You've probably seen these things lurking on worksheets. Maybe you've even tackled one yourself, staring down what felt like a cryptic crossword puzzle. Welcome to Chem Worksheet 19-2, where the stars of the show are these inseparable duos.
So, what are these pairs, really? Imagine two buddies who are practically glued at the hip. One is the acid, and the other is its ever-so-loyal base. They're like a dynamic duo, always doing something together.
This isn't just any old friendship, mind you. It's a very specific kind of relationship. They're connected by a simple act: the transfer of a proton. Yes, a tiny little proton is the secret handshake that binds them.
Let's break it down, super simply. When an acid loses a proton, what's left behind is its conjugate base. Poof! It's like the acid did a magic trick and shed a part of itself. And the base? It just patiently waited for this to happen.
On the flip side, when a base grabs a proton, it transforms into its conjugate acid. It's like the base got a power-up! Suddenly, it's got an extra hydrogen ion chilling with it.
Think of it like this: an acid is a generous donor. It's like that friend who's always willing to share their snacks. A base, on the other hand, is a keen collector. It's always looking for something to add to its collection, and that proton is a prime collectible!
Now, about that worksheet. You’re probably presented with a chemical equation. Your mission, should you choose to accept it, is to spot these pairs. It’s like a game of chemical hide-and-seek. Some are easy to find; others are a bit more elusive.
Let's say you see a molecule labeled as an acid. You then look at the products side of the equation. What’s there that looks almost like the original acid, but is missing just one little proton? Bingo! That's its conjugate base.
Similarly, if you see a base on the reactant side, scan the products. Is there something there that looks like it just absorbed a proton? That’s its conjugate acid. It's basically the base's upgraded version.
This is where the "unpopular opinion" comes in, my friends. While everyone else is stressing about complex formulas and balancing equations, I'm over here just appreciating the sheer elegance of these pairs. They're the unsung heroes of acid-base chemistry!
It’s not about memorizing a million different compounds. It’s about understanding the relationship. It’s like understanding how a parent and child are related, but in the chemical world. One gives, one receives, and they're forever linked.
Sometimes, the same molecule can act as an acid in one reaction and a base in another. These are the true chameleons of the chemical kingdom! They're adaptable, versatile, and probably have a more exciting social life than most of us.
When you’re working through Chem Worksheet 19-2, don’t let the fancy names intimidate you. Just remember the proton. That single, solitary proton is the key to unlocking the entire mystery.

Think of an acid as someone who hands over a valuable item. That item is the proton. What they’re left holding after the hand-off is their conjugate base. It’s like the item they no longer possess.
And the base? It’s the eager recipient. It’s waiting with open arms (or whatever bases have) to catch that proton. Once it’s got it, it becomes the conjugate acid. It’s now the proton-holding champion.
It’s really that straightforward. The "acid" and its "conjugate base" differ by one proton. The "base" and its "conjugate acid" also differ by one proton. Simple, right?
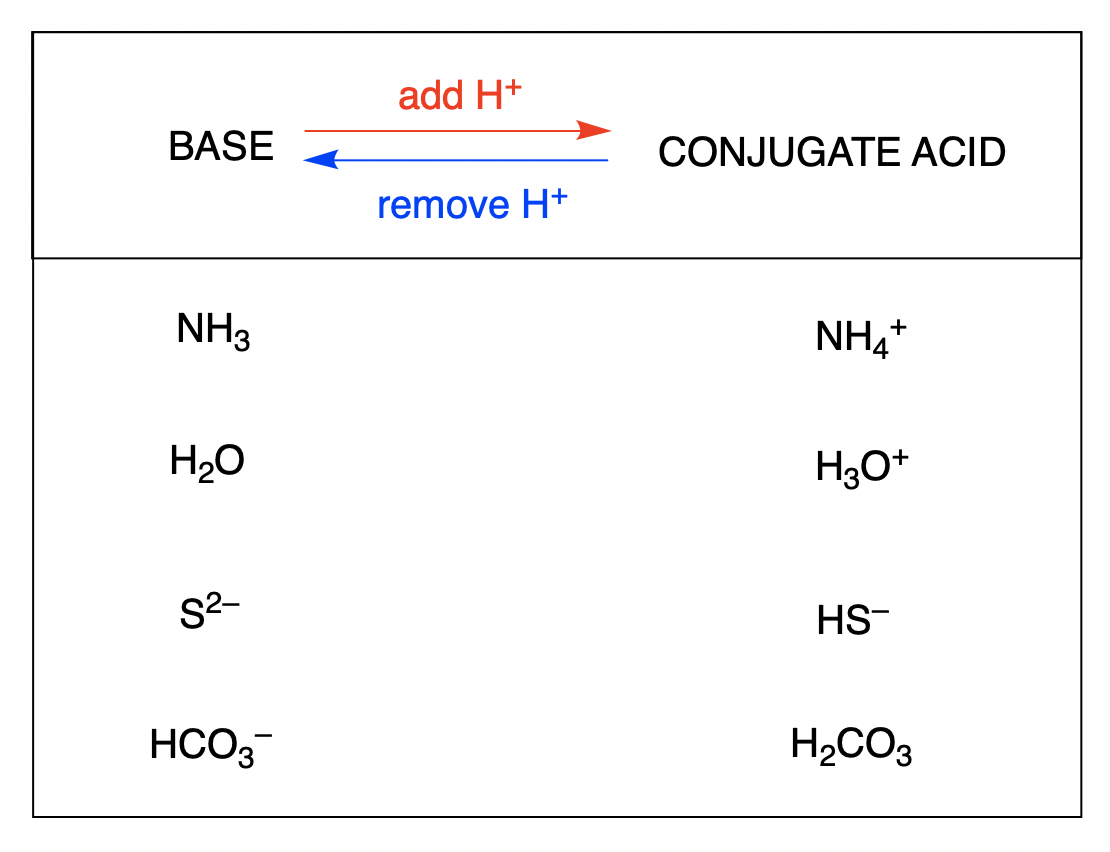
Let’s take a classic example. Water (H₂O) can be a bit of a wild card. When it acts as an acid, it donates a proton. What’s left? Hydroxide (OH⁻). So, H₂O and OH⁻ are a conjugate acid-base pair.
But wait, there’s more! Water can also act as a base. If it accepts a proton, what do you get? Hydronium (H₃O⁺). So, H₃O⁺ and H₂O are also a conjugate acid-base pair. See? Versatile!
It's like having two sides to a coin. You can't have one without the other in this context. They're two parts of the same chemical story.
Many worksheets will present you with a list of substances. Your job is to find the match for each one. It's like playing a game of "go fish" with protons.
You might see something like ammonia (NH₃). If it's acting as a base, it'll grab a proton to become ammonium (NH₄⁺). So, NH₃ and NH₄⁺ are a pair.
Or you might see something like hydrochloric acid (HCl). It's a classic acid. It’ll readily give up its proton, leaving behind chloride ion (Cl⁻). Thus, HCl and Cl⁻ are a pair.
The beauty of it is that once you grasp the proton-transfer concept, these problems become less about memorization and more about logical deduction. You're becoming a chemical detective!
And here's another thought: these pairs are everywhere! They're not just confined to your textbook or that slightly intimidating worksheet. They're the silent workers in many chemical processes, from biological systems to industrial applications.
So, the next time you encounter Chem Worksheet 19-2, don’t sigh. Don’t groan. Instead, put on your thinking cap and embrace the proton-transfer dance. See if you can’t spot those inseparable buddies, the conjugate acid-base pairs.
It's a fundamental concept, and understanding it makes so many other areas of chemistry click into place. Plus, it gives you something cool to talk about at parties. "Did you know that water can be both an acid and a base?" Boom. Instant fascinating conversation starter.
So, let's give a little cheer for these proton-exchanging pals. They’re the reason why so much of chemistry makes sense. They’re the true MVPs of the acid-base world.
Embrace the pairs. Love the pairs. And may your proton transfers be ever in your favor as you tackle those worksheets!
My Unpopular Opinion:
Honestly, I think conjugate acid-base pairs are way cooler than they get credit for. They’re like the ultimate chemistry bromance/sisterhood. They’re inseparable, defined by a single shared (or once-shared) proton. It’s the purest form of chemical connection, really. Forget complicated bonds; just give me a good old proton swap any day!