Hey there, science explorer! Ever feel like chemistry is all about complicated structures and big, scary words? Well, buckle up, because today we're going to dive into something super chill: a compound that throws the whole "closed chain" idea right out the window! Think of it like breaking free from a restrictive diet – this molecule is all about freedom and being wonderfully… open.
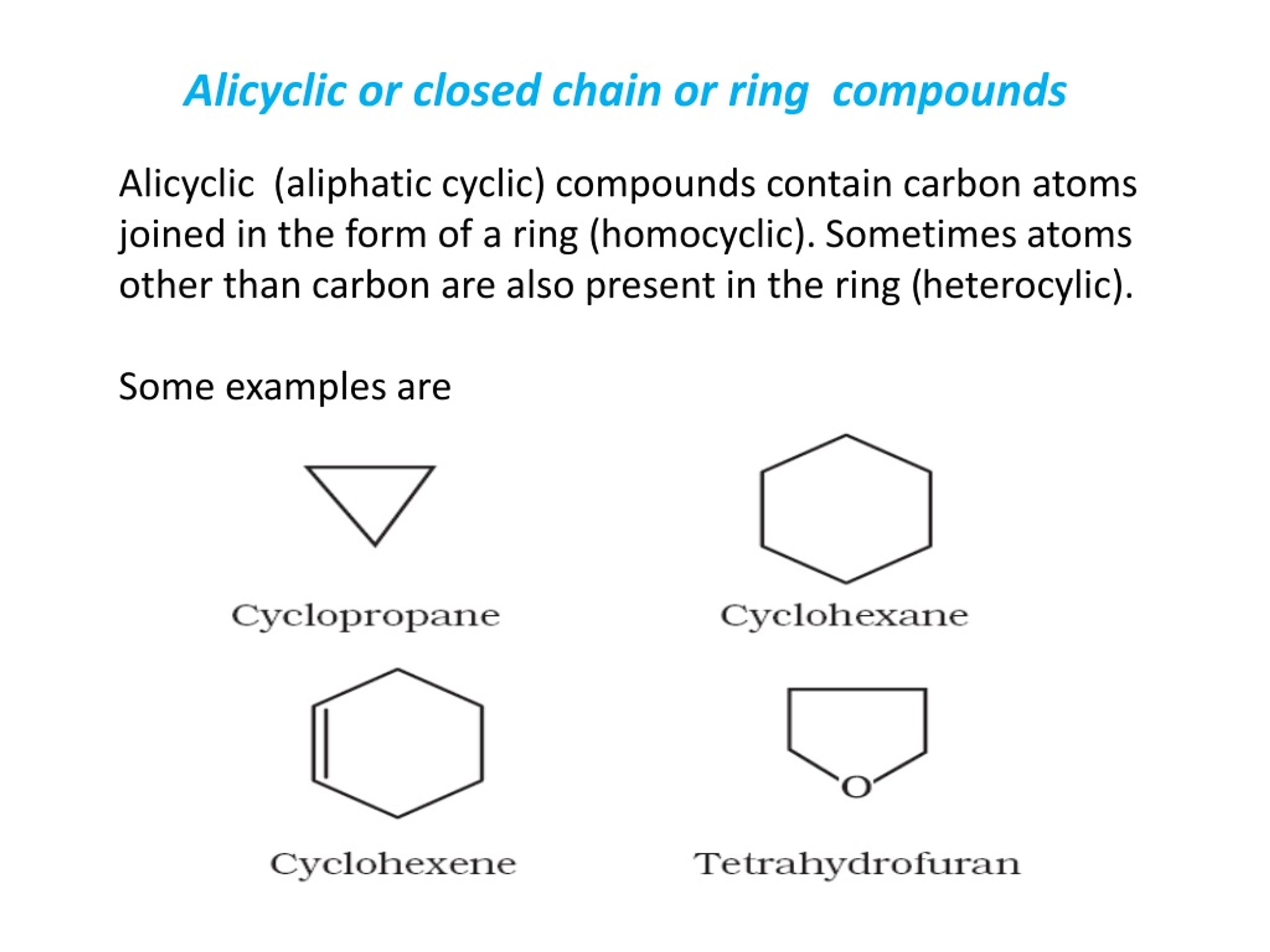
So, what in the world is a "closed chain" anyway? Imagine a necklace where the ends are perfectly linked together, forming a perfect circle. In chemistry, this is like a ring structure. Think of benzene (that classic hexagonal one!) or cyclohexane (its super-happy, slightly bent cousin). These guys are the epitome of closed chains. They’ve got their atoms all neatly arranged in a loop, no loose ends, no escape routes. They’re the ultimate in molecular commitment!
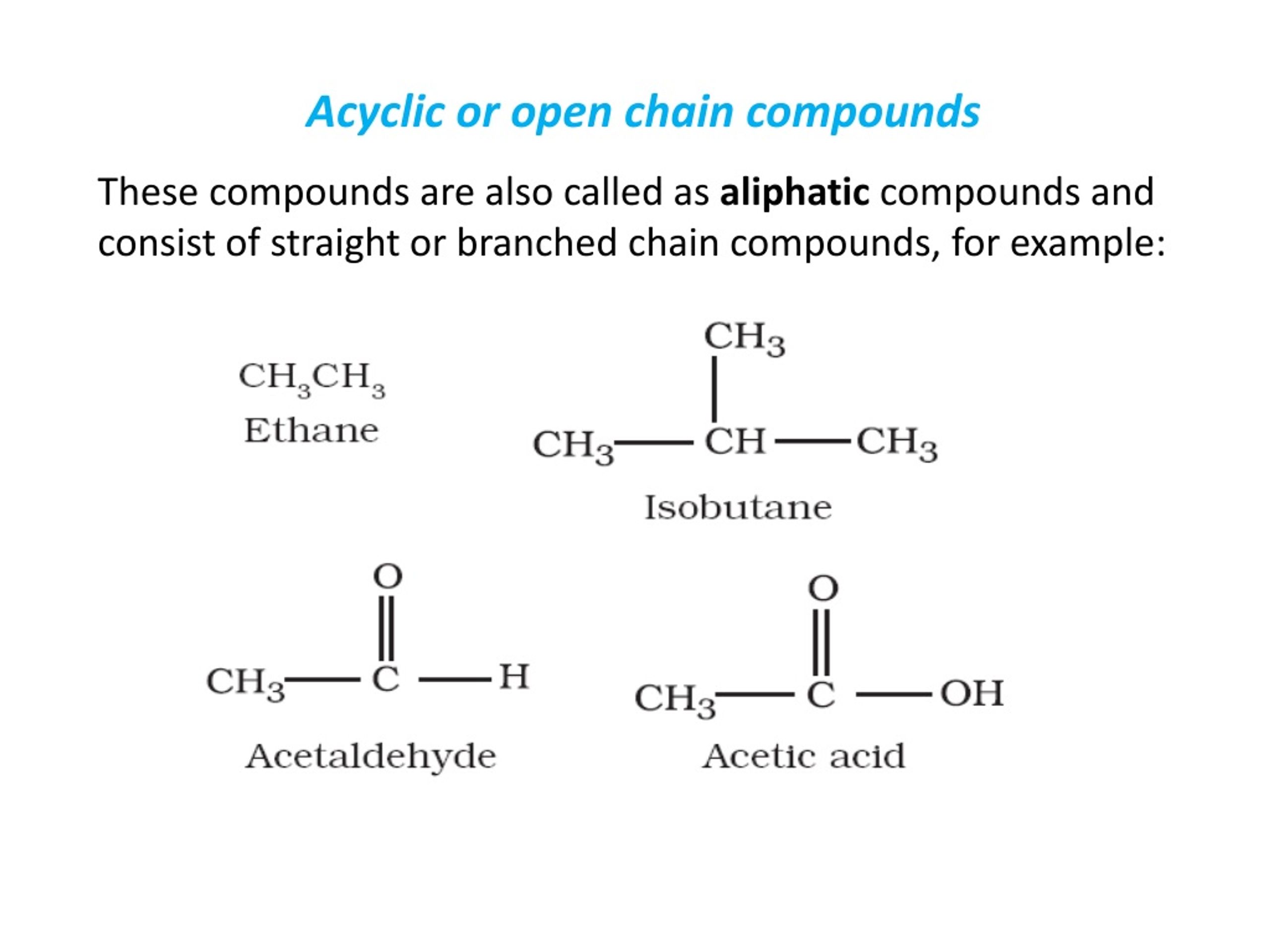
But here’s the fun part: some molecules just aren’t into that kind of commitment. They’re more like, "Eh, let's just hang out in a line." These are our open-chain compounds, also known as aliphatic compounds. Fancy word, I know! But all it really means is that they have a chain of atoms that doesn’t loop back on itself. It’s like a winding road instead of a race track. Much more potential for adventure, right?
Let’s talk about the simplest of the simple. Have you ever heard of methane? No, not the stuff that makes cows famous (though it's related!). This is CH4. One little carbon atom chilling with four hydrogen atoms. It’s the toddler of the organic world – uncomplicated, essential, and kind of the building block for everything else. And guess what? It’s definitely not a closed chain. It's a single carbon atom with arms outstretched to hydrogen buddies. So un-chained!
Then we move up the ladder. We’ve got ethane (C2H6). Two carbons linked together, with hydrogens happily attached to their sides. Think of it like two friends holding hands, and then each of them has other friends holding their hands. It’s still a line, not a circle. You can keep going! Propane (C3H8) – three carbons in a row, like a little conga line of carbon atoms. And butane (C4H10), the stuff in your lighter (don't play with that, kids!). All these are just straight or slightly wiggly lines of carbon atoms, decorated with hydrogen. They’re the ultimate proof that you don’t need a fancy loop to be a molecule.
These open-chain compounds are also called alkanes if they only have single bonds between the carbon atoms. Single bonds are like the sturdy handshakes of the molecular world. They’re strong and stable. So, methane, ethane, propane, butane… they’re all alkanes, and they’re all gloriously open-chain. No hoops to jump through, literally!
But wait, there's more! What if those carbon atoms decide to get a little frisky and form double or triple bonds? We’re still talking open chains here, folks! Take ethene (C2H4). It has a double bond between the two carbons. It’s like they’re holding hands really tightly! This extra bit of "stickiness" makes ethene behave a bit differently, but it's still a straight-up line of atoms. It's like a dance move where two people are holding hands extra tight – still part of the line, just with a bit more flair.
And then there's ethyne (C2H2), also known as acetylene. This one has a triple bond! Whoa nelly! The two carbon atoms are basically glued together with three bonds. Talk about a strong connection! But again, still an open chain. They're just really, really close. You can imagine them as two gymnasts performing an incredible synchronized routine, but they're still on the floor, not orbiting each other in a circle. It's all about that linear arrangement.
These compounds with double and triple bonds are called alkenes and alkynes, respectively. And you guessed it, they’re all members of the open-chain club. They might have different personalities due to those extra bonds, but their structural spirit is all about going in a straight (or slightly bent) line.
Now, you might be thinking, "Okay, that's all well and good, but what's the big deal?" Well, the big deal is that these open-chain structures are everywhere. They are the backbone of so many things we interact with every single day. Think about fuels: gasoline, propane for your BBQ, natural gas – these are all largely made up of open-chain hydrocarbons. So, the next time you’re grilling burgers, you can thank the wonderfully un-chained molecules for that delicious char!

And it’s not just fuels. Plastics! So many plastics are built from repeating units of open-chain molecules. That water bottle you’re drinking from? Likely a product of molecules that refused to be tied down in a ring. It's pretty wild to think that something as simple as a straight line of atoms can lead to something as complex and useful as a synthetic material.
Even in our own bodies, open-chain compounds play starring roles. Think about the building blocks of life, like amino acids. They have a central carbon atom, but the chains extending from it are open. These are the guys that build proteins, which do… well, pretty much everything in your body. They help you digest food, build muscle, even think! So, in a way, your very existence is a testament to the power of open chains.
Let's talk about a few more examples just to really drive this home. Have you ever heard of alcohols? Like ethanol (the kind in your celebratory drink – responsibly, of course!). Ethanol is basically ethane with one of its hydrogen atoms swapped out for an -OH group. So, C2H5OH. Still an open chain! It’s like one of the hydrogen atoms decided to bring a new friend along for the ride, but the overall structure remained linear. It’s the molecular equivalent of a small, friendly addition to the family gathering.
What about aldehydes and ketones? These are super important classes of organic compounds. Think of formaldehyde (a bit notorious, but a key building block) or acetone (nail polish remover – smells pretty intense, right?). These molecules have a carbonyl group (a carbon double-bonded to an oxygen, C=O). While this group adds a lot of reactivity and interesting properties, the carbon chain attached to it is still typically open. So, even with that flashy double bond to oxygen, the core structure can be nice and linear. They’re like a molecule wearing a very stylish, reactive accessory.

And then there are the carboxylic acids. These are molecules that have that carbonyl group plus an -OH group attached to the same carbon atom. Think of acetic acid, the key ingredient in vinegar. It’s got that acidic tang because of that special functional group. But the carbon chain before it? You guessed it – usually an open chain. So, even with these functional groups that make them react in certain ways, the underlying arrangement can still be the wonderfully straightforward open chain.
The beauty of these open-chain compounds lies in their versatility. Because they don't have the constraints of a ring, they can twist, turn, and extend in all sorts of ways. This allows for a massive diversity of structures and properties. Imagine a painter with a limited palette of only circular shapes versus a painter with a full spectrum of lines, curves, and angles. The latter can create so much more! The open-chain molecules are the artists of the chemical world, painting complex and beautiful structures.
Consider something like polyethylene, the most common plastic. It’s made by linking together thousands of ethylene (ethene) units. This creates a ridiculously long chain of carbon atoms, often thousands upon thousands of them, all in a row. It’s like a super-mega conga line of carbon atoms, and that's what gives it its flexibility and strength as a material. No loops, just one massive, un-chained highway of atoms.
Even when we're talking about molecules that can form rings, many of their important forms are open-chain. For example, a sugar molecule like glucose can exist as an open chain or a ring. But its open-chain form is crucial for how it's used in metabolism. So, even if a molecule has the potential to form a ring, its open-chain form is often just as, if not more, important.

So, to recap, a compound that does not contain a closed chain is essentially any molecule where the atoms are arranged in a linear or branched fashion, rather than forming a loop. These are your alkanes, alkenes, alkynes, and many of the functionalized compounds derived from them, like alcohols, aldehydes, ketones, and carboxylic acids. They are the unpretentious workhorses of the chemical world.
They might not have the elegant symmetry of a benzene ring, or the compact nature of a cyclic molecule, but they have a freedom and flexibility that makes them incredibly important. They are the foundation of so many materials, fuels, and biological processes. They are the straight roads that lead to countless destinations.
And here’s the really cool part: this simple concept of an "open chain" is a gateway to understanding so much more in chemistry. It’s a reminder that not every structure has to be complicated. Sometimes, the most fundamental and widespread molecules are the ones that simply refused to be confined. They chose to be open, to be flexible, to be available for all sorts of connections and transformations.
So, the next time you see a molecule, don’t just look for rings. Appreciate the beauty of the straight lines, the gentle curves, the branching paths. Celebrate the compounds that dare to be open. Because in that openness, there’s a whole universe of possibilities, just waiting to be discovered. And that, my friends, is something pretty amazing to smile about!




