Imagine you’ve got a favorite recipe, something that’s been passed down through generations. You know it by heart, the way the flour feels, the exact moment the dough is perfectly smooth, the comforting aroma that fills your kitchen. Now, imagine you could give that recipe a special, invisible fingerprint. That’s kind of what we did with benzoic acid. It’s not a cake, but it’s a super important ingredient in a lot of things we use every day, from your grandma’s famous pickles to that squeaky clean feeling you get from some soaps.
But how do we know it’s really benzoic acid, and not something else that’s snuck in? Well, chemists have a neat trick up their sleeves, and it’s called Infrared (IR) Spectroscopy. Think of it like giving each molecule its own unique musical note. When you shine a special kind of light – infrared light, which we can’t even see – on our benzoic acid, it starts to wiggle and jiggle in a very specific way. Each of these wiggles absorbs a tiny bit of the infrared light at a particular “note” or frequency.
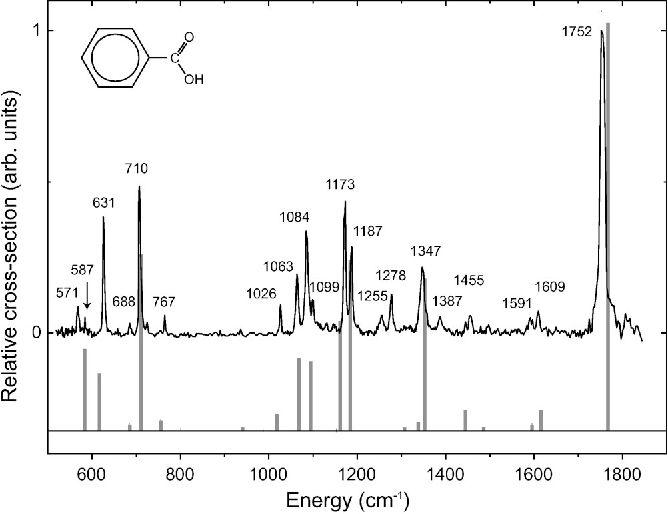
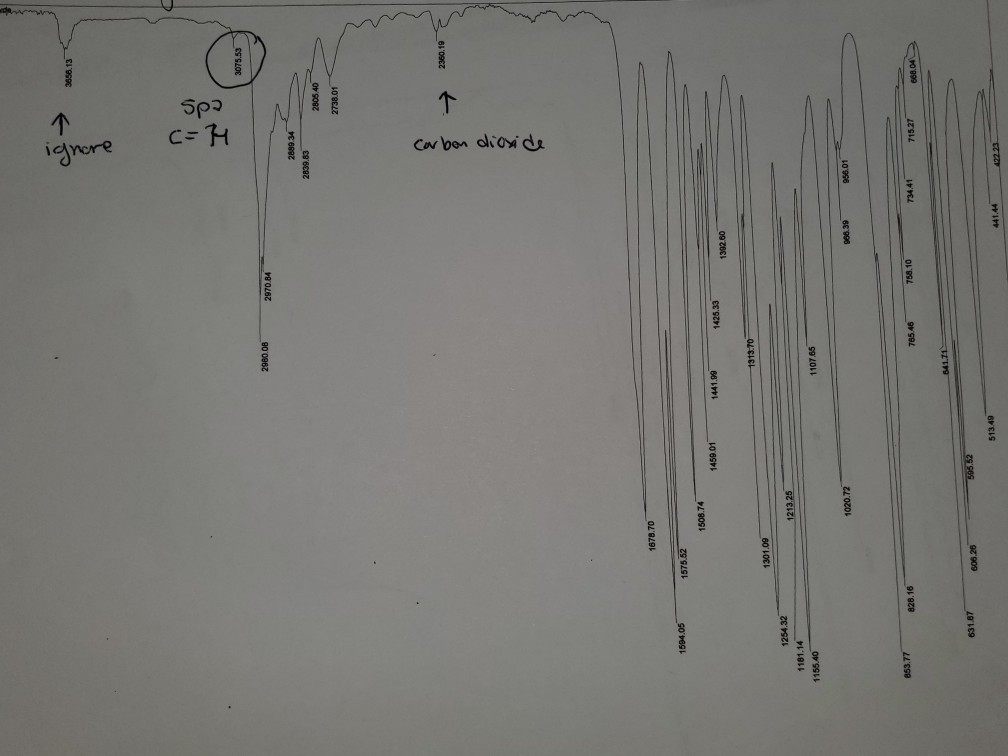
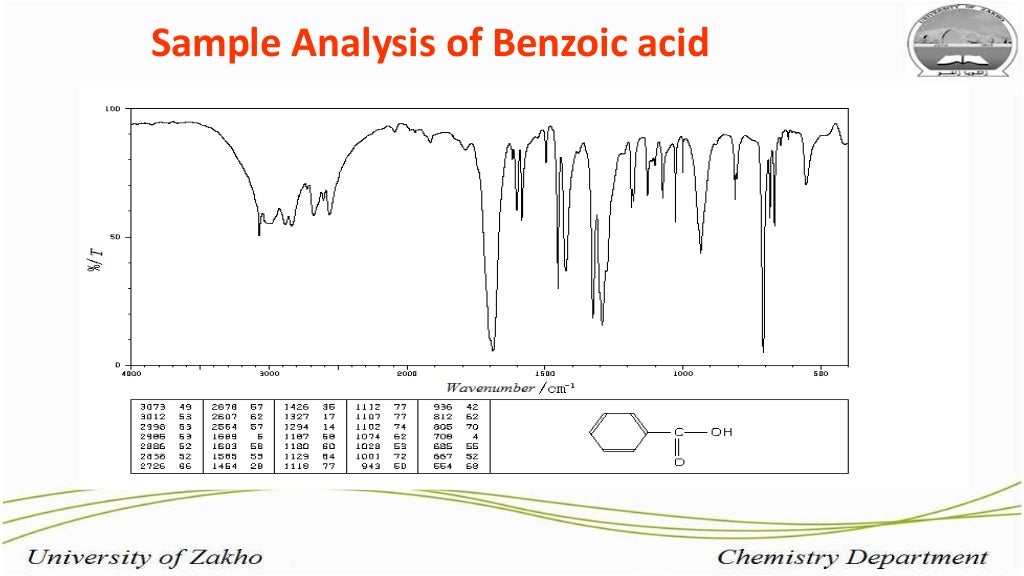
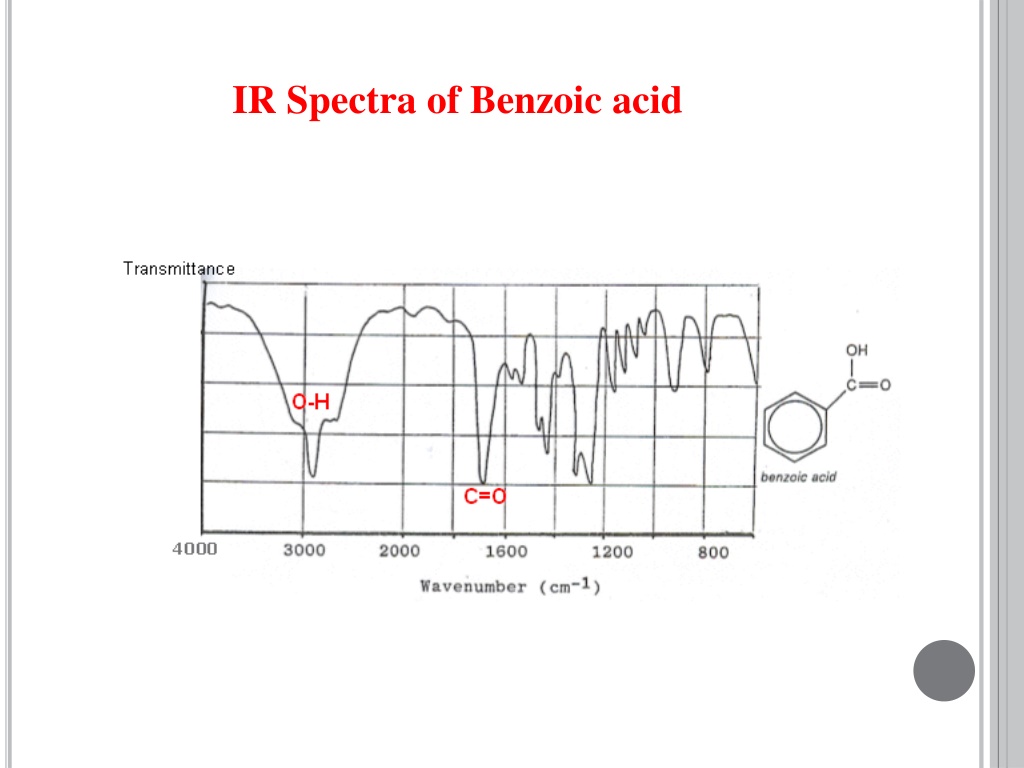
Now, here’s where the fun starts. We don’t just look at the wiggles. We actually record them. It’s like taking a photograph of the molecule’s dance. This record is called an IR Spectrum. For benzoic acid, this spectrum looks like a bunch of little dips and valleys, almost like a jagged mountain range drawn by a child. Each dip tells us something important about the molecule. It’s like a secret code that only the scientists (and the benzoic acid itself!) truly understand.
Let’s peek at some of the star performers in this spectral show. There’s a big, broad shoulder of a dip that usually sits around the 2500-3300 cm-1 mark. This one is a bit like a shy whisper from a special kind of hydrogen atom, one that’s hanging out with a very tight grip on an oxygen atom. It’s part of the carboxyl group (that’s the -COOH part of benzoic acid), and its presence is a big clue that we’re dealing with something acidic. It’s like the molecule is shyly clearing its throat before it really belts out its other tunes.
Then, we have another rather prominent dip, usually a bit sharper and found in the 1680-1725 cm-1 region. This one is the unmistakable “voice” of the carbonyl group, that’s the C=O part. It’s like the lead singer hitting a powerful note. This particular vibration is super important because it’s very characteristic of carboxylic acids like our friend benzoic acid. It’s a confident declaration: “Yes, I am indeed a carboxylic acid!”

But benzoic acid isn’t just about the -COOH group. It also has a ring of six carbon atoms, a benzene ring, which is where the "benzoic" part of its name comes from. This ring has its own unique set of wiggles. You’ll often see some strong, crisp dips in the 1450-1600 cm-1 area, which are the tell-tale signs of the carbon-carbon double bonds within that aromatic ring. These are like the rhythmic beat of the molecule’s heart, keeping everything together.
And if you look further down the spectrum, towards the higher numbers (like around 690-900 cm-1), you’ll find some more finger-like patterns. These are often associated with the way the hydrogen atoms are attached to the benzene ring. Each molecule has its own special arrangement, and these patterns are like the unique signature of how those hydrogens are seated. It’s like looking at a crowd of people – they’re all human, but each one has a slightly different way of standing or gesturing.
So, what’s the heartwarming or humorous part of all this? Well, imagine a scientist, maybe after a long day of experiments, looking at this spectrum. It’s not just a bunch of lines on a graph. It’s a confirmation. It’s like getting a postcard from a molecule, saying, “Yep, it’s me! And I’m doing just fine!” It brings a sense of certainty and satisfaction. It’s the invisible assurance that the substance you’re working with is exactly what you expect it to be. It’s like knowing your favorite comfort food is made with the genuine, secret ingredient, and not some imposter.
“This spectrum is essentially the molecule’s autobiography, written in the language of light and vibration.”
For something as common and useful as benzoic acid, having such a clear and distinct spectral fingerprint is a big deal. It means we can confidently use it in everything from preserving food to making useful chemicals. It’s the quiet, unsung hero that ensures quality and reliability in so many of the products we rely on. So, the next time you see “benzoic acid” on a label, remember its invisible dance, its unique musical notes, and the special fingerprint that IR Spectroscopy reveals. It’s a little bit of scientific magic that keeps the world running smoothly, one molecule at a time.