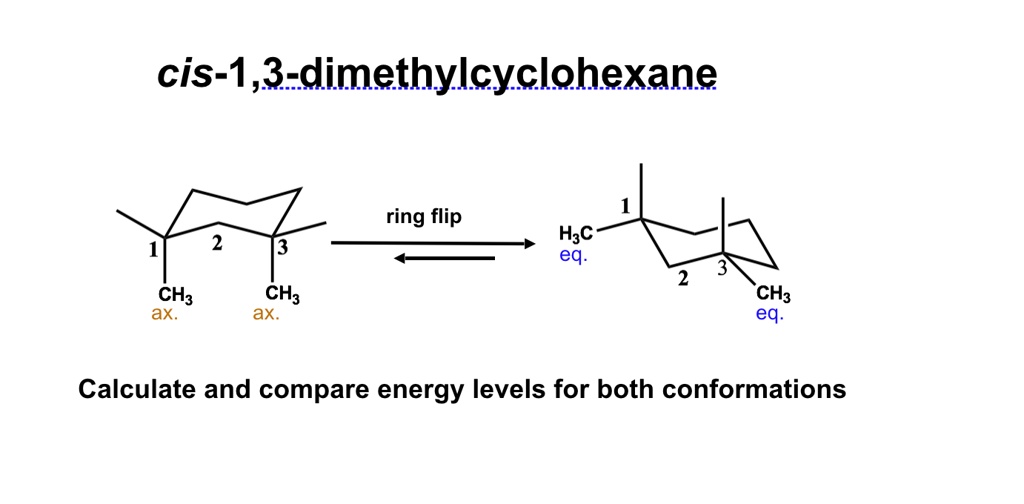
Imagine your favorite comfort food. Now, imagine that comfort food has a secret life, a hidden world of twisty, turny arrangements that makes it… well, even more comforting. That’s kind of what we’re diving into today, but instead of pizza or cookies, we’re talking about a rather charming little molecule called cis 1,3 dimethylcyclohexane.
Now, don't let the name scare you! Think of it like this: you have a ring of six carbon atoms, like a tiny, six-person bicycle wheel. We've stuck two little "methyl" groups onto this wheel, which are basically just tiny carbon-and-hydrogen arms. And the "cis" part? That’s the fun bit – it means these arms are on the same side of the wheel, like two friends giving each other a high-five over the handlebars.
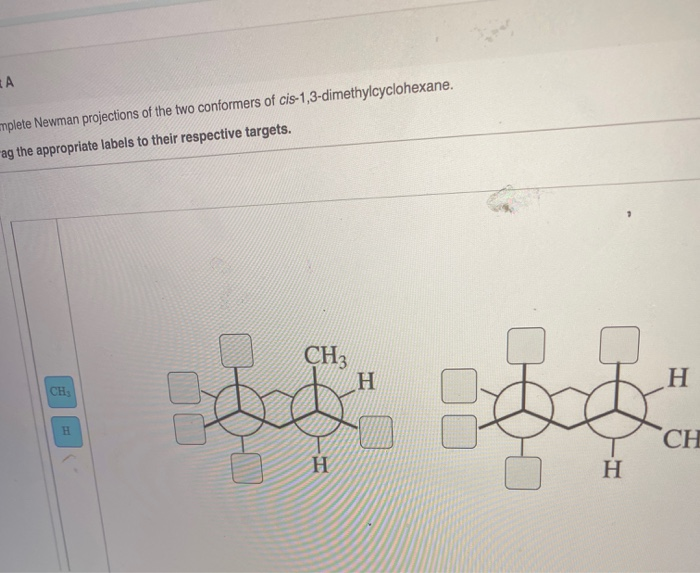
So, where does the "Newman Projection" come in? It's like taking a snapshot of this tiny molecule from a specific angle. Imagine you're looking straight down one of the carbon-carbon bonds. You see one carbon as a dot (the front one), and the other as a circle behind it.
And then, sprouting from these two carbons, are the other bits of the molecule. It's a bit like a weird, abstract drawing, but it tells us so much about how these atoms are arranged in space.
Let’s focus on our cis 1,3 dimethylcyclohexane. Remember those two "arms" (the methyl groups)? Because they're "cis," they're pointing in the same direction relative to the ring.
When we draw its Newman Projection, we get a specific pattern. One of those methyl groups will be pointing "up" (or "down," depending on your viewpoint) relative to the ring, and the other will be in a similar general direction.
The surprising part? Even though it's the same molecule, there are slightly different ways it can arrange itself to get the most comfortable. Think of it like a cat curling up in a sunbeam. It might stretch out a bit, or tuck itself in, but it's always trying to find that perfectly cozy position.

For our cis 1,3 dimethylcyclohexane, these different cozy arrangements are called "conformations." And the Newman Projection helps us visualize them.
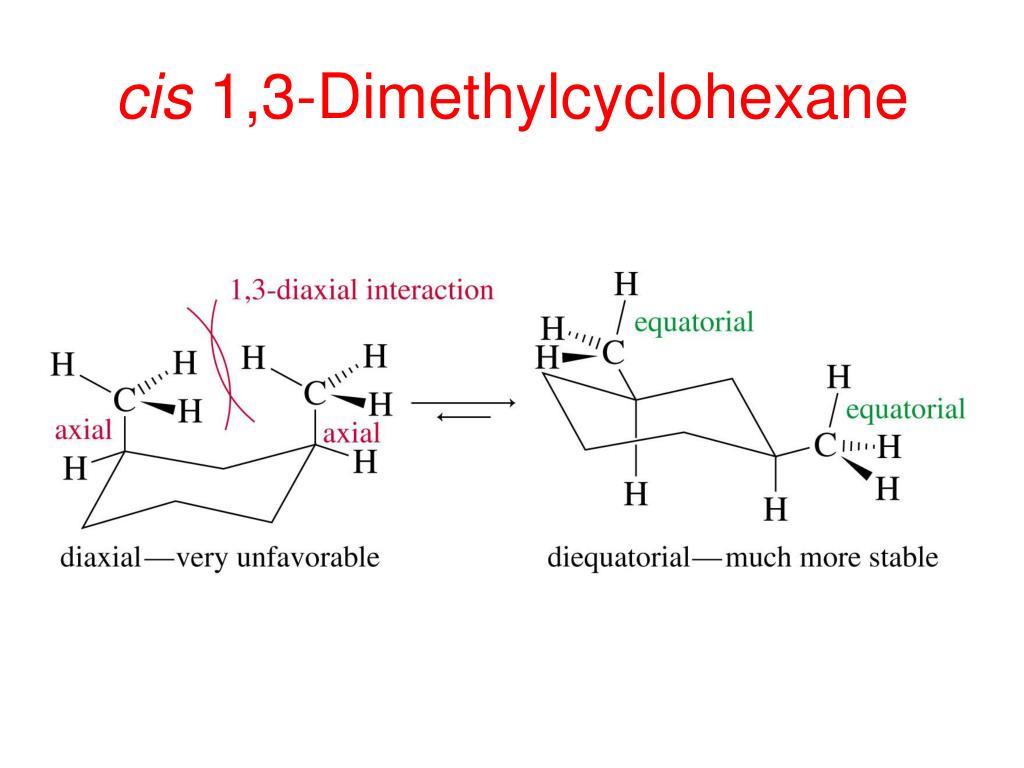
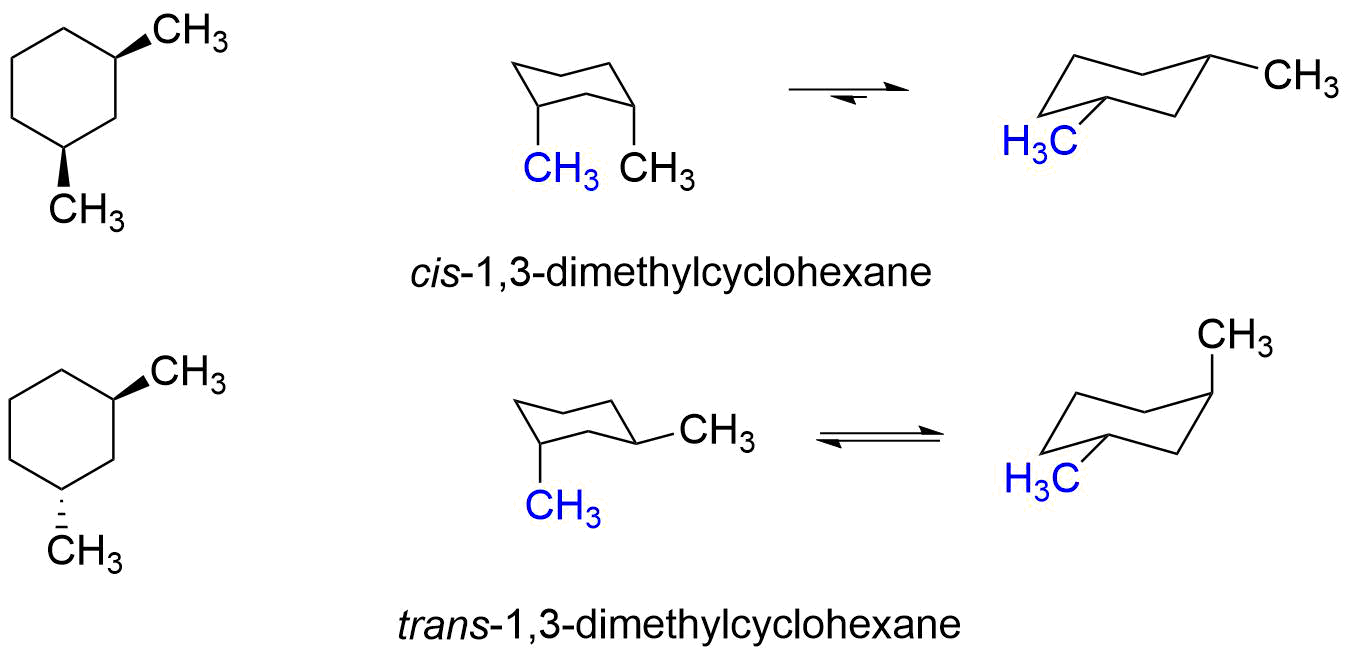
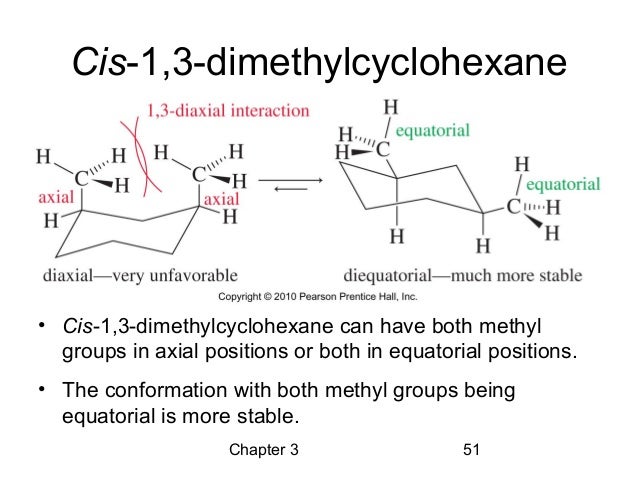
One common comfy pose for our molecule is where both methyl groups are in what we call "equatorial" positions. Imagine the bicycle wheel again. The equatorial positions are like sitting on the outside edge of the wheel, sort of hugging the rim.
When both methyl groups are equatorial, they're pretty far apart. This is a happy, stable arrangement because they aren't bumping into each other. It's like having plenty of personal space at a crowded party.
Another arrangement, less comfy but still possible, is when one methyl group is equatorial and the other is "axial." Think of axial positions as being like standing straight up from the center of the wheel, pointing towards the sky or the ground.
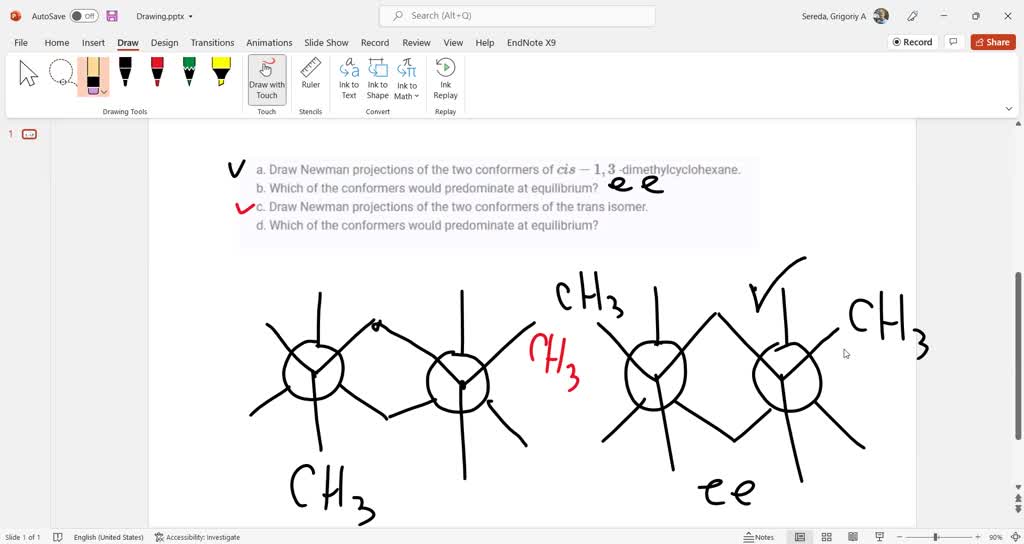
In this case, the two methyl groups are a bit closer. They might give each other a gentle nudge, making this arrangement a tiny bit less preferred. It's like having to stand a little too close to someone you don't know that well.
And then, there’s the least comfy arrangement. This is where both methyl groups are axial. Now they're really crammed together, practically giving each other a full-on hug.
This arrangement is like being stuck in a tiny elevator with someone you really don't want to be stuck with. There's a lot of "steric hindrance," which is just a fancy way of saying "things are getting too crowded."
So, why is this even remotely fun or heartwarming? Because it shows us that even the tiniest building blocks of our world are constantly seeking comfort and stability. Our cis 1,3 dimethylcyclohexane, in its own molecular way, is trying to find the best possible way to exist.
It's a little bit like us, isn't it? We adjust our posture, we shift around, we find the perfect pillow – all to feel more at ease. This molecule is doing the same thing, just on a scale we can't even see.
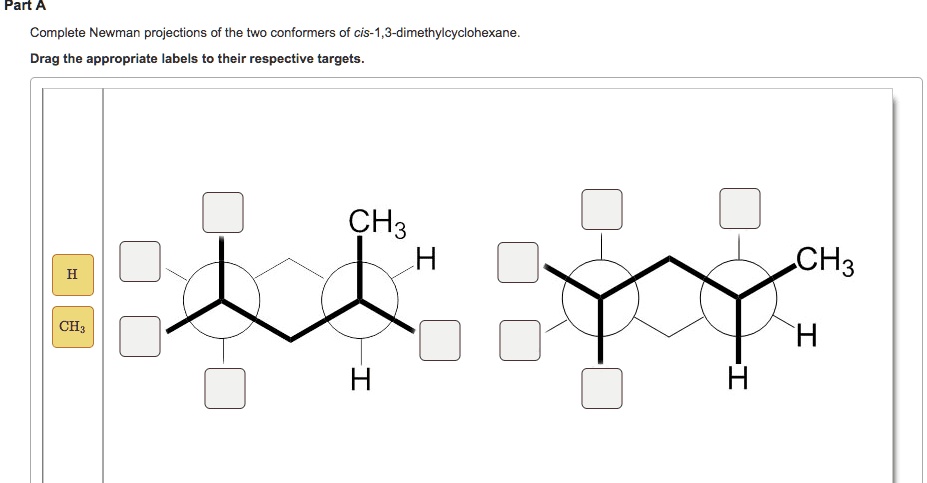
The beauty of the Newman Projection is that it takes this invisible dance and makes it visible. It's like having a secret window into the molecular world, where we can see these tiny shapes and their preferences.
Think of the cis 1,3 dimethylcyclohexane as a tiny, six-wheeled performer, constantly adjusting its pose for the audience (which, in this case, is the universe itself). The Newman Projection is the stage lighting, highlighting its most graceful moves.
And the "cis" part? That's the special bond between two methyl groups, ensuring they start off facing the same way, setting the stage for their little molecular ballet. It’s a cooperative effort, a shared decision to be on the same side.
When we see the Newman Projection, we're not just looking at dots and circles. We're seeing a story of how these atoms arrange themselves to minimize discomfort and maximize their peaceful existence. It's a quiet, chemical harmony.
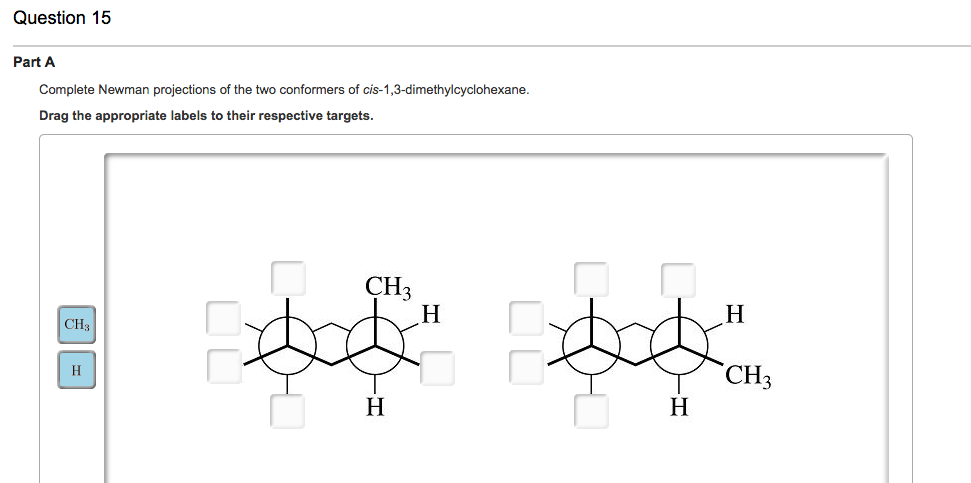
The most common Newman Projection for cis 1,3 dimethylcyclohexane will show you the most stable arrangement. It's the one where the methyl groups are doing a little jig, one equatorial, one axial, but not the super-crowded all-axial situation. It's a compromise, a balanced act.
Even the less stable arrangements have their charm. They show us that molecules, like us, can sometimes be in slightly awkward positions, but they still keep going. They adapt.
So, the next time you hear about a molecule like cis 1,3 dimethylcyclohexane, don't just think of it as a string of letters and numbers. Think of it as a tiny, intricate dancer, using its Newman Projection to show the world its elegant (and sometimes slightly cramped) moves. It’s a little bit of molecular magic, right there for us to see.
And the fact that we can use these simple drawings to understand such complex interactions? That's pretty amazing, isn't it? It’s like having a decoder ring for the universe.
This tiny molecule, with its "cis" high-five and its quest for the comfiest conformation, is a gentle reminder that even in the smallest corners of existence, there's a constant, subtle striving for balance and well-being. It’s a heartwarming thought, if you ask me.