Ever wondered about the tiny, invisible building blocks that make up everything around us? Atoms are pretty amazing, and learning about them can be surprisingly fun! Today, we're going to dip our toes into the fascinating world of chlorine, a common element that has a cool little secret: it actually comes in two naturally occurring flavors, called isotopes. It might sound a bit technical, but understanding this simple fact can add a whole new layer of appreciation for the chemistry happening all around us.
So, why should you care about chlorine's isotopes? For beginners, it's a gentle introduction to the idea that elements aren't always just one thing. Think of it like different kinds of apples – they're all apples, but some are sweeter, some are tarter, and they might have slightly different colors or textures. Chlorine isotopes are similar. For families, it's a great way to spark curiosity in kids. You can talk about how even something as ordinary as the chlorine in swimming pools has this hidden variation. For hobbyists, especially those interested in chemistry experiments or even just understanding the materials they use, knowing about isotopes can offer a deeper insight into how things work.
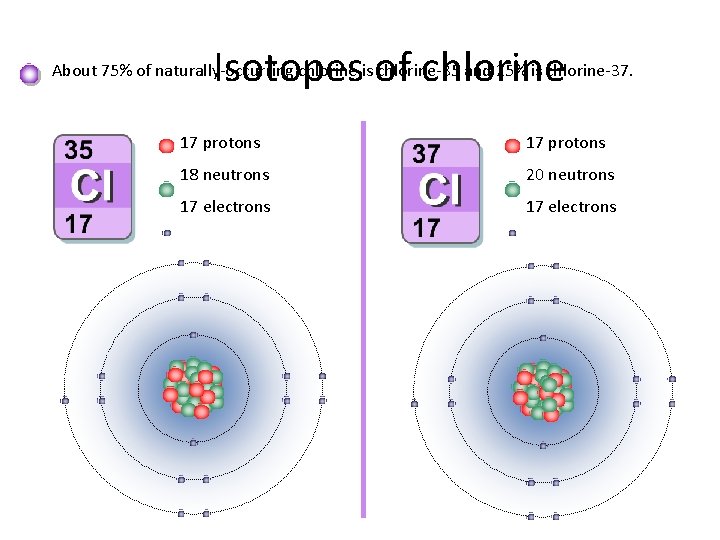
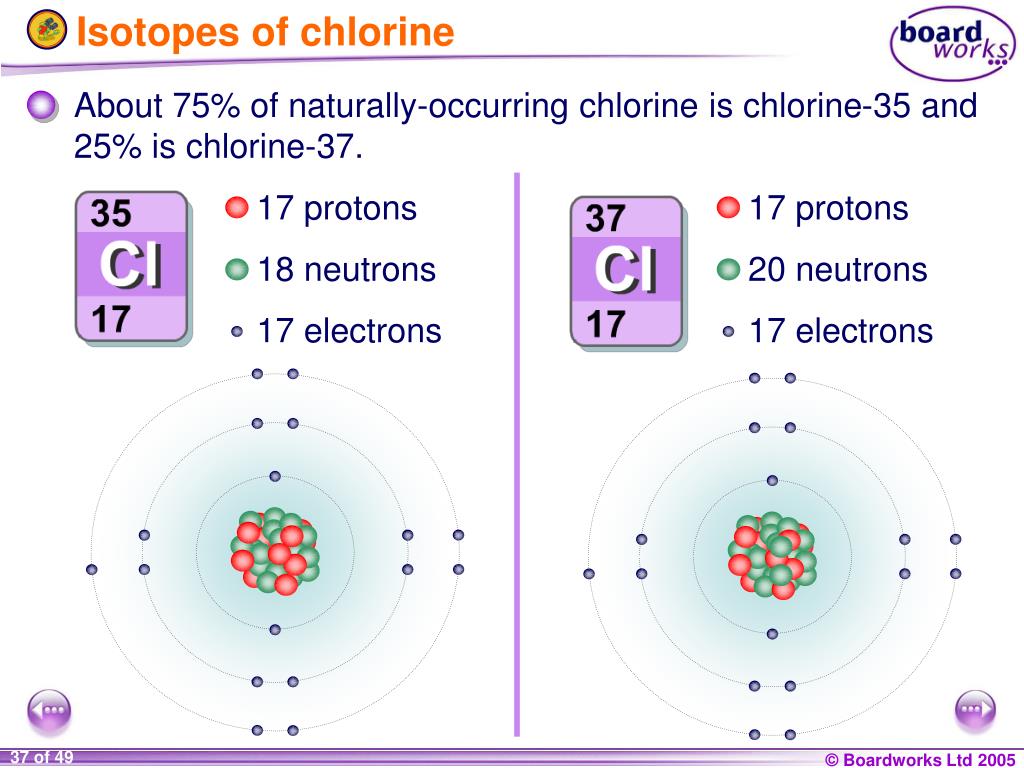
Let's get a little more specific. Chlorine, the element, is represented by the symbol 'Cl'. When we talk about isotopes, we're referring to atoms of the same element that have a different number of neutrons in their nucleus. Think of neutrons as the "extra weight" in the atom's core. Chlorine's two main naturally occurring isotopes are Chlorine-35 and Chlorine-37. The number after the dash tells you the total number of protons and neutrons in the atom's nucleus. So, Chlorine-35 has 17 protons (that's what makes it chlorine) and 18 neutrons, while Chlorine-37 has 17 protons and 20 neutrons. This slight difference in weight is the primary distinction!
What are some real-world connections? While you won't see different types of chlorine floating around in the pool, this isotopic difference is incredibly important in scientific fields. For instance, in environmental studies, scientists use the ratios of different chlorine isotopes to track the movement and history of pollutants. In forensic science, isotopic analysis can help identify the origin of certain materials. Even in understanding ancient climates, scientists look at isotopes! So, while it's a microscopic difference, it has macroscopic implications.
Getting started with this concept is easier than you think! You don't need a lab coat. A great first step is to simply read more about elements and isotopes online or in a beginner's science book. You can search for "what are isotopes" and "uses of chlorine isotopes" to find accessible articles and videos. Perhaps try a simple activity with your family: find different types of fruits and talk about how they are all fruit, but have unique characteristics. This can be a fun analogy for isotopes. If you're a hobbyist, consider looking into introductory chemistry kits that might explain atomic structure in more detail, or even just exploring online resources for chemistry enthusiasts.
Learning about the two naturally occurring isotopes of chlorine might seem like a small detail, but it opens up a whole world of understanding about the fundamental nature of matter. It’s a reminder that even the most common elements have hidden complexities and fascinating variations, making the study of chemistry a truly rewarding and enjoyable journey.