Imagine you're baking your absolutely favorite cookies. You’ve got the recipe right there, promising pure deliciousness. It calls for 2 cups of flour, 1 cup of sugar, and 3 eggs. You grab your ingredients, ready to whip up a masterpiece!
Now, what if you only put in 1 cup of flour? Your cookies would probably turn out a bit… sad. Maybe flat, maybe crumbly, definitely not the chewy, golden goodness you dreamed of. It just wouldn't work, would it?
Well, guess what? The universe is a bit like that cookie recipe. When things happen on a super tiny, invisible level – we're talking about atoms and molecules – they also need to follow a strict rulebook. And this rulebook says: everything has to balance out!
The Cosmic Balancing Act
Think of it like a cosmic game of LEGOs. You have a pile of red bricks and a pile of blue bricks. When you’re building something, and the instructions say you need 4 red bricks and 2 blue bricks, you can't just use 3 red ones and call it a day. The structure would be wobbly, right?
That's exactly what happens in chemistry when we talk about chemical reactions. It's like a tiny construction project happening all around us, all the time. Things are breaking apart, rearranging, and building themselves into something new.
And just like your cookie recipe, the "ingredients" (which are called reactants) have to perfectly match the "finished product" (which are called products). You can't magically create new LEGO bricks out of thin air, and you can't just make atoms disappear!
Meet the Mighty __________!
Okay, so there’s this super important concept in chemistry that makes sure our LEGO constructions and cookie recipes make sense. It’s called the Law of Conservation of Mass. Fancy name, right? But all it really means is that you can't create or destroy matter. It just changes its outfit!
So, if you start with 2 hydrogen atoms and 1 oxygen atom, and they decide to team up, what do you get? You get water! But here’s the catch: the number of each type of atom you started with has to be exactly the same as the number you end up with. No exceptions!

It’s like having a perfectly organized playroom. Every single toy you put in the toy box at the beginning of playtime has to be accounted for when it's time to clean up. You can’t lose a teddy bear or find a brand new dinosaur that wasn’t there before!
The Drama of Imbalance
Now, let's have some fun with what happens when things are not balanced. Imagine you’re trying to assemble a tiny robot. The instructions clearly state you need 1 head, 2 arms, and 1 body. You’ve got all the parts!
But then, oops! You accidentally drop one of the arms. Now you have a robot with only one arm. It might still look like a robot, but it's incomplete, right? It’s not the robot the instructions promised.
In chemistry, an unbalanced equation is like that one-armed robot. It’s a jumbled mess, a scientific oopsie. It tells us something went wrong in the recipe, and the atoms didn't play fair.
When Atoms Play Hide-and-Seek
Think about making a simple salt and water mixture. You mix salt (sodium chloride) with water (H₂O). On the surface, it seems straightforward. But on the atomic level, things are happening!

The sodium (Na) atoms and the chlorine (Cl) atoms in the salt, and the hydrogen (H) and oxygen (O) atoms in the water, they're all interacting. If our chemical "recipe" isn't balanced, it’s like some atoms just decided to take a vacation and never came back. Or even worse, they multiplied out of nowhere!
It’s like trying to count your fingers and toes, but some of them have mysteriously vanished, or you suddenly sprout an extra thumb. It's just not right, and it makes the whole process feel a bit… chaotic.
Enter the Equation Stabilizers!
So, how do scientists make sure everything stays in line? They use something called a balanced chemical equation. This is the universal "balance sheet" for atoms. It’s like the ultimate inventory checklist!
When we write out a chemical reaction, we’re essentially listing the ingredients and the results. A balanced equation shows us that the number of each type of atom on the "ingredient" side (reactants) is exactly the same as the number of that type of atom on the "results" side (products).
It’s like having a super-organized librarian who makes sure every book that goes into the library at the beginning of the day is still accounted for at the end. No magic book disappearances or spontaneous arrivals!

The Art of Counting Atoms
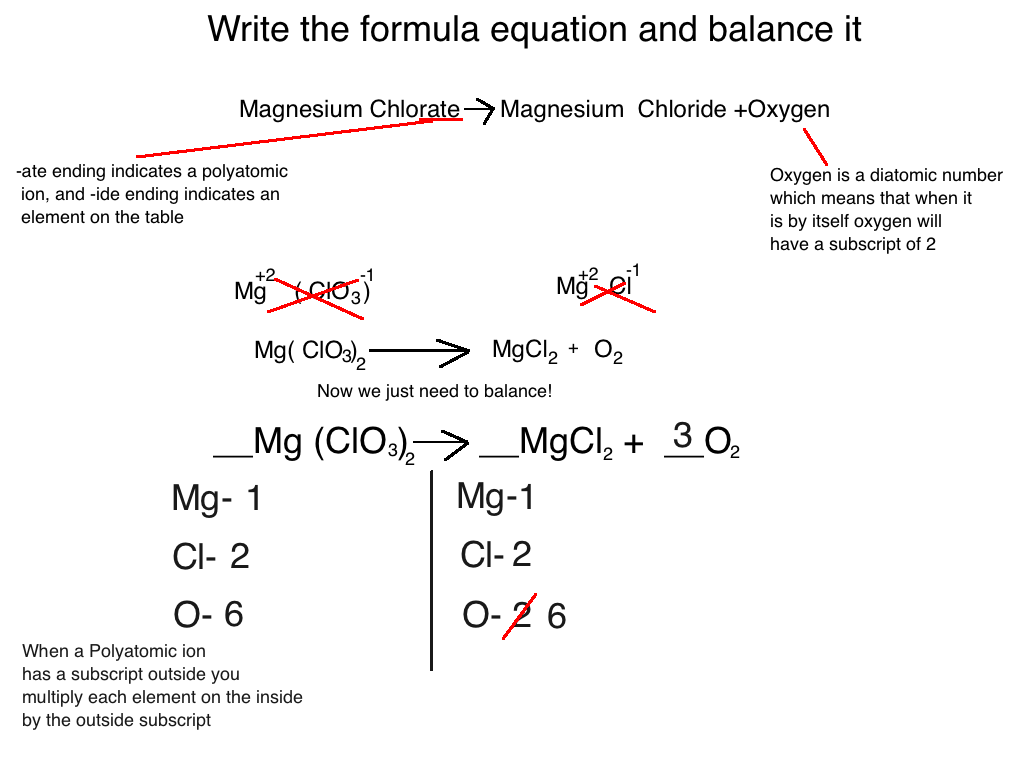
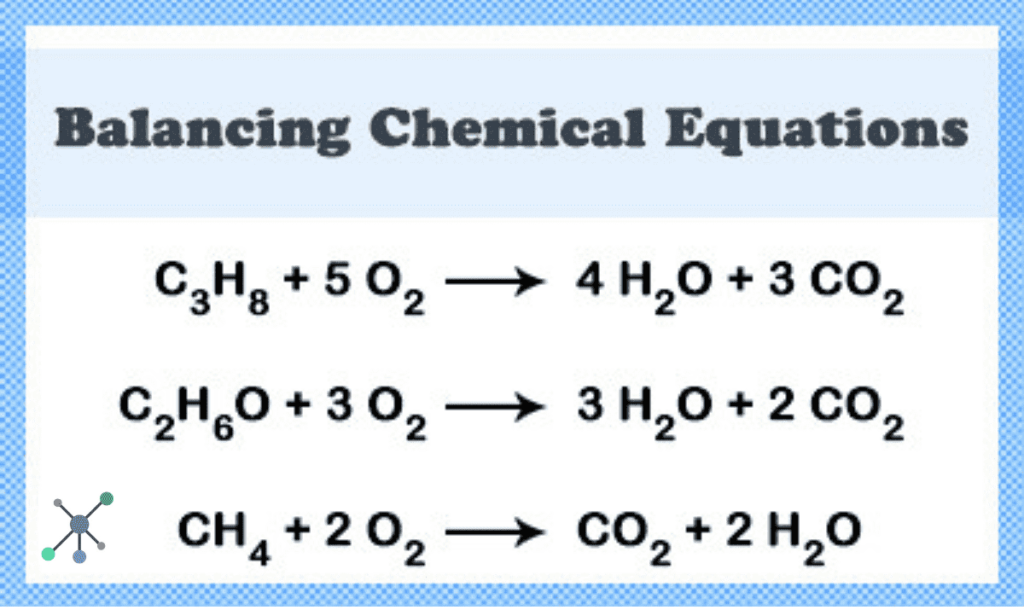
Let’s take the formation of water. The recipe is: Hydrogen + Oxygen → Water. In symbols, it looks like H₂ + O₂ → H₂O.
On the left side, we have two H atoms and two O atoms. On the right side, we only have two H atoms and one O atom. Uh oh! One oxygen atom is missing its buddy!
To fix this, we add numbers in front of the chemical formulas. This is like saying, "Hey, we need more of that ingredient!" We’re not changing the type of thing, just how much of it we’re using. It’s like telling the cookie recipe, "Actually, let's use two eggs instead of one to make sure everything works perfectly."
So, we adjust it to 2H₂ + O₂ → 2H₂O. Now, let’s count! On the left, we have 2 x 2 = 4 hydrogen atoms and 2 oxygen atoms. On the right, we have 2 x 2 = 4 hydrogen atoms and 2 x 1 = 2 oxygen atoms. Ta-da! Everything matches!
Why This Matters (Besides Atomic Peace)
Why is this whole balancing act so important? Well, for starters, it’s fundamental to understanding how the world works. Every single thing you see, touch, and even are, is a result of chemical reactions that follow these balancing rules.

From the food you eat to the air you breathe, and even the amazing technology that powers our lives, it all relies on balanced chemical equations. If these reactions weren't balanced, our entire universe would be a chaotic, unpredictable mess. Imagine trying to drive a car where the fuel randomly produced half the energy it was supposed to!
It’s also crucial for scientists. Whether they’re developing new medicines, creating eco-friendly materials, or exploring the vastness of space, accurate chemical equations are their roadmap. They need to know exactly what’s going in and what’s coming out to predict outcomes and ensure safety.
A Universe in Harmony
So, the next time you’re cooking, marveling at a sunset, or even just taking a deep breath, remember the incredible, silent work of balanced chemical equations. They are the invisible architects of our reality, ensuring that every atom plays its part and that the universe remains in perfect, harmonious balance.
It's a beautiful, intricate dance of atoms, and when they're all accounted for, the results are nothing short of miraculous. It’s a reminder that even the smallest things, when organized correctly, can create something truly spectacular. Cheers to balanced equations and a world that just… works!