Okay, so picture this: My first ever attempt at baking sourdough. I’d seen all these gorgeous, bubbly loaves online, heard tales of its magical tang, and thought, "How hard can it be?" Turns out, very hard, if you don't understand a thing called equilibrium. My starter looked sad, the dough wouldn't rise, and the final product was… well, let's just say it was more like a brick than a loaf. I spent days fussing over it, feeding it, poking it, muttering sweet nothings to it, all while it stubbornly refused to do what it was supposed to do. It was frustrating! And then it hit me. It wasn't about brute force; it was about finding that sweet spot, that balance. Sound familiar? Because that, my friends, is basically what Chapter 18, "Chemical Equilibrium," is all about.
You’re probably here because you’ve just tackled that chapter, wrestled with the concepts, and now you’re staring down the barrel of the answer key. Maybe you’re feeling a bit like I did with my sourdough – a mix of hope and mild panic. Will the answers make sense? Will they clarify the fog, or just add another layer of confusion? Let’s dive in and see if we can untangle this whole equilibrium thing, one question at a time, shall we?
So, What's the Big Deal with Equilibrium Anyway?
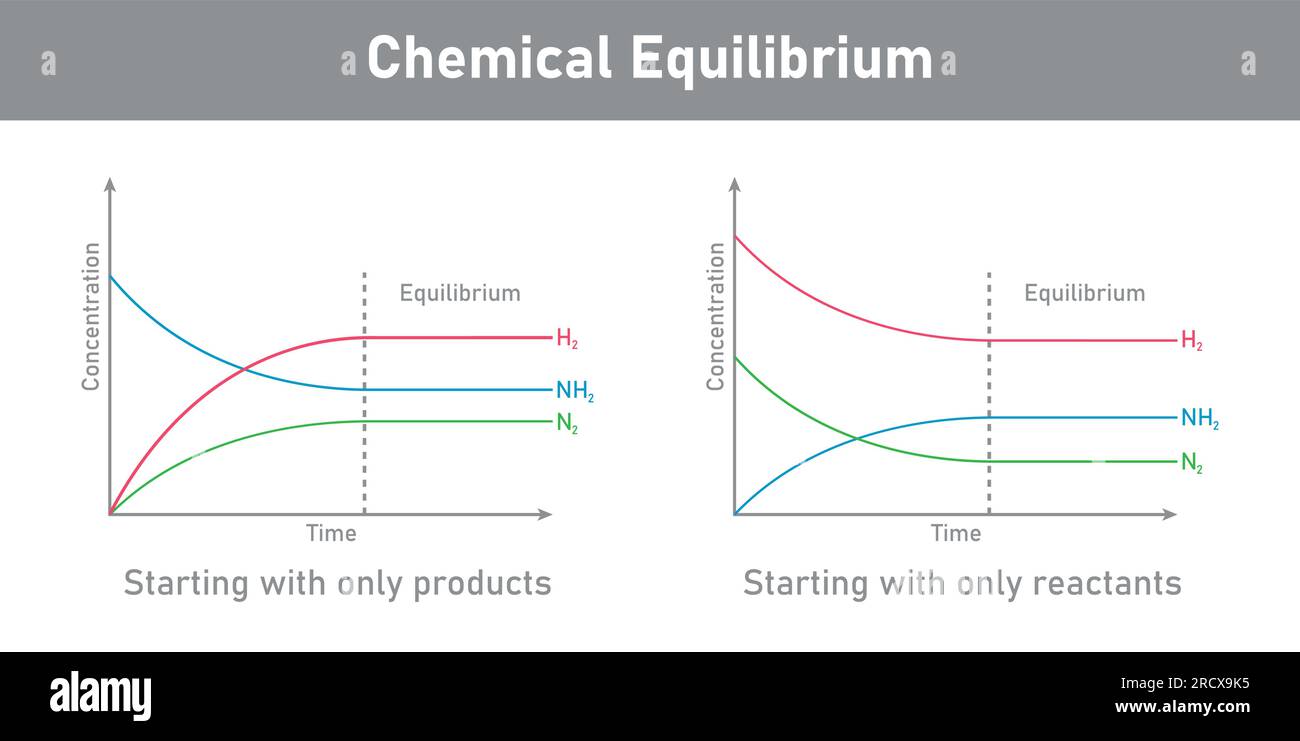
In a nutshell, chemical equilibrium is when a reversible reaction has reached a point where the rate of the forward reaction (reactants turning into products) is exactly equal to the rate of the reverse reaction (products turning back into reactants). It's not that the reaction stops; oh no, it's still humming along, just like a busy city street where people are constantly entering and exiting buildings, but the overall number of people on the street remains constant. It’s a state of dynamic balance. Pretty neat, right?
Think of it like this: Imagine you have two rooms connected by a door, and people are moving back and forth. At first, everyone might be in Room A. But then, some people start drifting into Room B. As Room B gets more crowded, more people might start moving back to Room A. Equilibrium is reached when the number of people moving from A to B each minute is the same as the number of people moving from B to A each minute. The total number of people in each room stays the same, even though individuals are still moving.
This concept is HUGE in chemistry because so many reactions don't just go to completion. They reach this equilibrium point, and understanding where that point lies tells us a lot about how much product we can expect to get. And that, my chemistry comrades, is where the answer key to Chapter 18 becomes your new best friend (or at least a very helpful study buddy).
The Key Players: Kc and Kp
Now, the chapter probably introduced you to these two little letters that hold a lot of power: Kc and Kp. These are your equilibrium constants. They’re basically numbers that tell you the ratio of products to reactants at equilibrium. A big Kc or Kp means you’ll have a lot of product at equilibrium – your reaction is leaning heavily towards making stuff. A small Kc or Kp means you’ll have more reactants hanging around – the reaction isn’t super keen on making a ton of product.

Kc is for concentrations (usually in molarity), and Kp is for partial pressures (if your reactants/products are gases). They’re related, of course, because pressure and concentration of gases are linked. The answer key is going to be full of calculations involving these, so getting a solid grip on what they represent is crucial. Don't just memorize the formulas; try to understand the meaning behind that number. Is it a big number? Celebrate! Your reaction is working hard for you.
Decoding the Answer Key: Common Pitfalls and "Aha!" Moments
Alright, let's get down to brass tacks. You’ve got the questions, you’ve got the answers, and you’re trying to connect the dots. Here are some common areas where students (myself included!) often stumble, and how the answer key might shed some light.
Calculating Equilibrium Concentrations/Pressures
This is often the meat and potatoes of equilibrium problems. You’ll be given initial amounts and then asked to find the amounts at equilibrium. This usually involves setting up an ICE table (Initial, Change, Equilibrium). If you’re looking at the answer key and it’s suddenly giving you numbers, and you’re thinking, "Where did that come from?", chances are they’re using an ICE table or a similar systematic approach. Make sure you’re comfortable with how the 'x' value (the change) is applied – it’s usually multiplied by the stoichiometric coefficient from the balanced equation. This is a critical step!
Sometimes, you might have to solve for 'x' using a quadratic equation. If the answer key shows a nice, clean number, and you got a messy decimal or two solutions, double-check your math. But don't be surprised if the "real world" answers involve some approximation or a calculator. It's all about finding that balance point, remember?

The Magic of Le Chatelier's Principle
Ah, Le Chatelier's Principle. The superhero of predicting shifts in equilibrium. If you add more reactant, the system shifts to use it up. If you increase the temperature, the system tries to cool down. If you increase pressure (for gases), the system tries to reduce it. The answer key will likely have questions testing your understanding of these shifts. When you look at the answers, ask yourself: Why did it shift that way? Which direction did it go (forward or reverse)? And why did that specific change cause that shift?
For example, if the question is about adding a catalyst, and the answer is "no change in equilibrium position," that’s your cue to remember that catalysts speed up both the forward and reverse reactions equally. They help you reach equilibrium faster, but they don’t change where equilibrium lies. This is a subtle but super important point!
What about heat? If a reaction is exothermic (releases heat, ΔH is negative), adding heat is like adding a reactant for the reverse reaction. The equilibrium will shift left, favoring reactants. If it's endothermic (absorbs heat, ΔH is positive), adding heat favors the forward reaction. The answer key’s responses will directly reflect these principles. Don't just accept the answer; try to articulate the reasoning behind it based on Le Chatelier.

Solubility Equilibrium and Ksp
If your chapter touched on solubility, you’ve probably met Ksp, the solubility product constant. This is equilibrium in action for sparingly soluble salts. It tells you the maximum concentration of ions that can exist in a saturated solution. When you see calculations involving Ksp in the answer key, remember that it’s the same principle: a balance between the solid salt and its dissolved ions.
For instance, if you’re asked if a precipitate will form when you mix two solutions, you’re essentially comparing the ion product (Qsp) with Ksp. If Qsp is greater than Ksp, the solution is oversaturated, and precipitation will occur to reach equilibrium. The answer key will guide you through these comparisons. It’s all about whether the ion concentration is too high to stay dissolved.
Beyond the Numbers: Conceptual Understanding
While the answer key is great for checking your calculations, don't neglect the conceptual questions. Sometimes, the most challenging parts are the "why" and "how" questions, not the "what is the number" ones.
For example, a question might ask about the significance of the equilibrium constant. The answer isn't just a number; it's about what that number tells you about the reaction's tendency to proceed. A very large K means the reaction is essentially "complete" from a practical standpoint. A very small K means the reaction barely happens in the forward direction. The answer key will provide the concise, correct explanation, but your job is to internalize it.

Similarly, understanding that equilibrium is a state, not a process that stops, is fundamental. The molecules are still zipping around, reacting, but the net effect is no change. This is why even though the rates are equal, the concentrations aren't necessarily 50/50. The equilibrium constant dictates the ratio of concentrations at that dynamic balance.
Making the Answer Key Work FOR You
So, how do you use this answer key effectively, and not just as a crutch? Here's my advice:
- Attempt the problem FIRST. Seriously, give it your best shot before peeking. Struggle is where learning happens.
- When you check your answer, don't just look at the final number. Trace the steps. If you got it wrong, figure out where you went wrong. Was it a math error? A conceptual misunderstanding of Le Chatelier's? An incorrect ICE table setup?
- If you got it right, can you explain why? If the answer key says "shifts left," can you explain why it shifted left based on the disturbance? This is the mark of true understanding.
- Identify patterns. Do you keep messing up calculations involving gases? Or are you struggling with identifying exothermic vs. endothermic reactions? Use the answer key to pinpoint your weak spots and focus your study there.
- Re-do problems. After you’ve understood the answers and the reasoning, try the problem again a day or two later without looking at your notes. See if you can solve it from scratch.
Chapter 18 can feel like a bit of a beast, I’ll admit. It combines math, concepts, and predictions. But the beauty of chemical equilibrium is its universality. It applies to so many processes, from industrial synthesis to the chemistry happening inside your own body. The answer key is your roadmap, your guide, and your sanity check.
So, take a deep breath. Go through those answers, not just to confirm correctness, but to understand the logic. Treat it like a puzzle, and with each solved problem, you’re getting closer to mastering this fascinating concept. And who knows, maybe your next baking project will be a sourdough success story. Just remember the balance!