Get ready for a little molecular magic! Today, we're diving into the fascinating world of Bromine Trifluoride, or as we affectionately call it, BrF3. Think of it as a super-powered, yet surprisingly polite, chemical adventurer ready to show off its awesome shapes. It’s not just some abstract concept; it's like a miniature cosmic dance happening on a tiny scale, and we’re about to get front-row seats!
Imagine you're a chef, and you have your main ingredient, Bromine (Br). This is our star player, the central atom around which all the action happens. It's like the celebrity chef in the kitchen, holding court and orchestrating the whole meal. BrF3 has one Bromine atom, and it's feeling a bit fancy, so it decides to invite some friends.
And who are these friends? They are three cheerful Fluorine (F) atoms! Think of them as eager sous chefs, ready to follow their leader's instructions. They're a bit smaller and a lot more energetic, always ready to jump in and help out. Together, they form the compound BrF3, a dynamic duo, or rather, a dynamic quartet in this case!
Now, when these atoms get together, they don't just huddle up randomly. Oh no, they have rules! The most fundamental rule is about how they arrange themselves in space. This is where we talk about Electron Geometry. It’s like figuring out how your kids arrange themselves in the living room when they're playing – there are only so many ways they can spread out comfortably.
For BrF3, the Bromine atom is the boss, and it has some things it needs to position. It's got its three Fluorine friends, but that's not all! Bromine also has a couple of "lonely hearts" – these are lone pairs of electrons. Imagine these lone pairs as shy, but important, guests who also need their own space to feel comfortable.
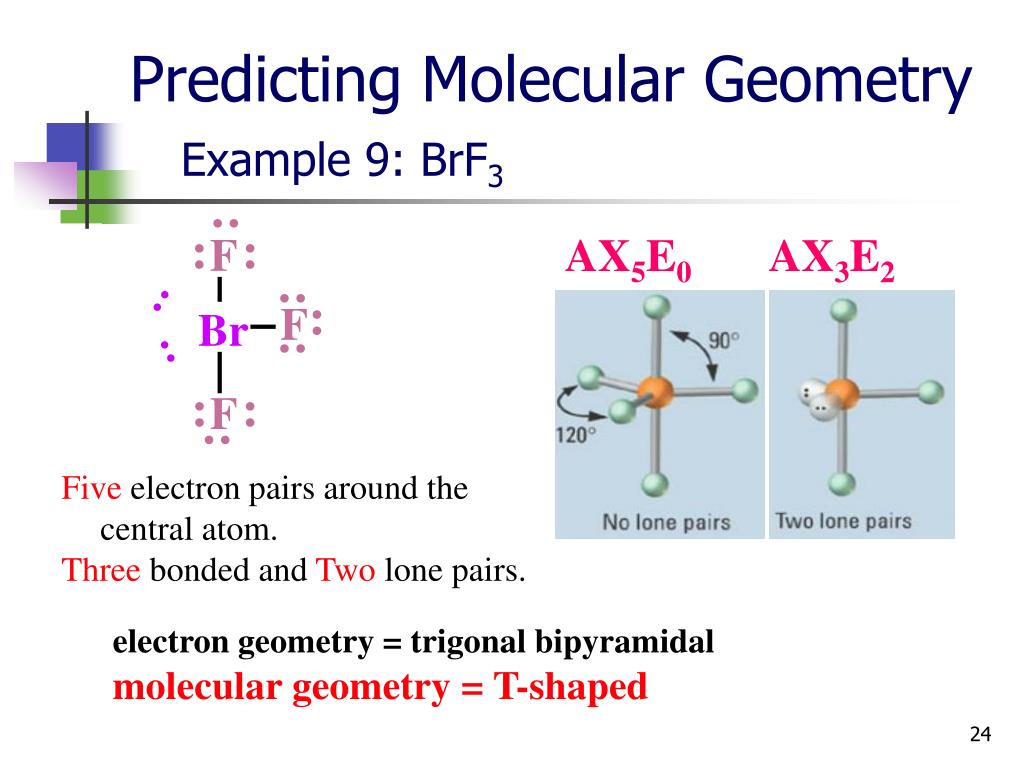
So, BrF3’s Bromine atom has a total of five electron groups to manage: three Fluorine atoms and two lone pairs. When you have five electron groups, the universe dictates they want to spread out as far as possible, like a group of balloons trying to avoid touching each other. This arrangement, where all electron groups (both bonding pairs and lone pairs) are trying to get away from each other, leads to a specific spatial layout.
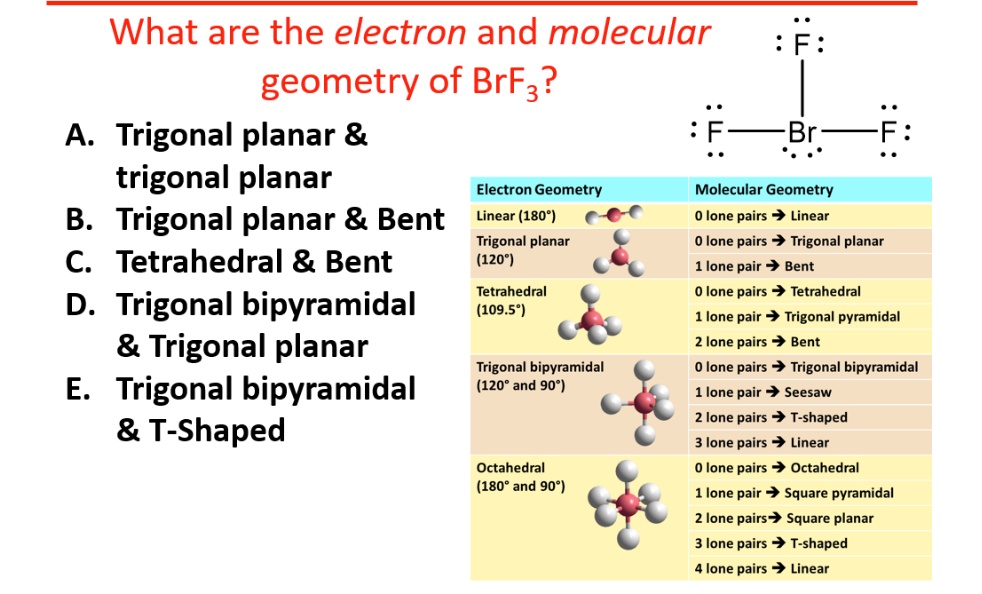
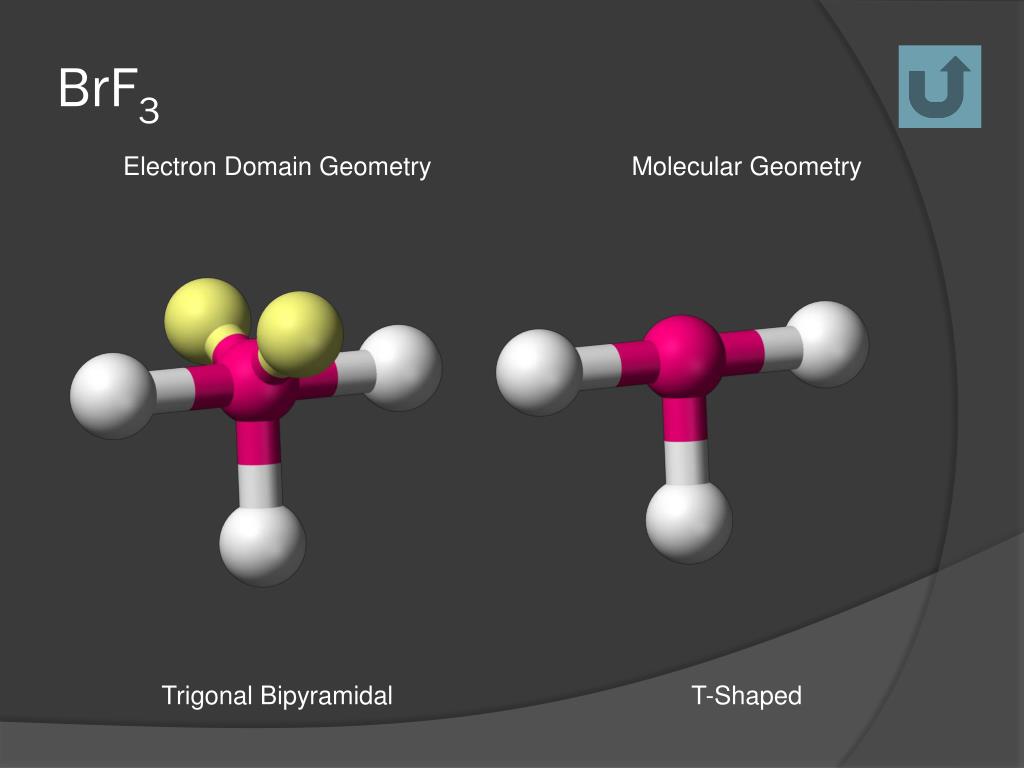
This fundamental layout, dictated by the desire of all electron clouds to be as far apart as possible, is called the electron geometry. For five electron groups, this preferred shape is a trigonal bipyramidal structure. Imagine a little pyramid with a triangle base, and then another pyramid stacked on top of that, with the Bromine atom right in the middle. It's like a futuristic spaceship base camp!
But here's the fun twist! While the electron geometry tells us where all the electron clouds want to hang out, we're often more interested in where the atoms themselves end up. This is where molecular geometry steps in, and it’s a whole different ballgame, like deciding where the actual players stand on the field, not just where the coach's instructions are written.
In BrF3, we have our central Bromine, and it's bonded to three Fluorine atoms. These are the actual pieces of furniture in our molecular room. However, those two shy lone pairs of electrons are still there, influencing the whole vibe!

Think of the lone pairs as bulky furniture that pushes the Fluorine atoms around a bit. Even though the electron groups want to be in a trigonal bipyramidal arrangement, the lone pairs are like overzealous interior decorators who rearrange the actual atoms. They take up a surprising amount of space and exert a stronger "push" on the bonding pairs.
So, while the electron geometry is trigonal bipyramidal (the ideal spacious layout for all five electron groups), the molecular geometry is not! Because those two lone pairs are so influential, they push the three Fluorine atoms into a specific, non-symmetrical arrangement.
Instead of a perfectly symmetrical shape, BrF3's Fluorine atoms end up arranged in a way that looks like a bent stick, or more formally, a T-shape. Imagine the Bromine atom as the corner of a giant T, and the three Fluorine atoms are the ends of the T, with one sticking straight up and two sticking out to the sides, slightly bent. It's like the T-shape wants to do a little jig!
So, to recap, the electron geometry is about the grand plan, the ideal spacing for all electrons. In BrF3's case, it's the ambitious trigonal bipyramidal plan. But then, the molecular geometry is the reality, the actual shape formed by the atoms, influenced by those bossy lone pairs. And for BrF3, that reality is a charming T-shape.
It’s like planning a party! The electron geometry is the blueprint for how many guests you can comfortably fit in your house (trigonal bipyramidal). But then, the actual arrangement of people, with some people clustering together and others trying to find their own corner, that’s the molecular geometry (T-shape)! The lone pairs are like the chatty groups who take up more room.
This difference between electron and molecular geometry is super important in chemistry. It helps us understand why molecules behave the way they do, how they interact with other molecules, and what their properties are. BrF3, with its T-shape, is a much more reactive and interesting molecule than if it were perfectly symmetrical!

The T-shape of BrF3 is what gives it its unique personality and its ability to be a powerful fluorinating agent. It's not just a random bend; it's a carefully crafted arrangement that makes it a formidable chemical force. Think of it as the elegant, yet slightly asymmetrical, dance move that makes it stand out!
So, the next time you hear about BrF3, remember its secret! It's got a grand electron geometry of trigonal bipyramidal, but it rocks a more distinctive T-shape molecular geometry, all thanks to those influential lone pairs. It’s a testament to how subtle differences in electron arrangement can lead to vastly different molecular personalities. How cool is that?
It just goes to show, even in the world of atoms and electrons, there's always a bit of playful chaos and unexpected charm. BrF3 is a fantastic example of this, proving that sometimes, the most interesting shapes are the ones that aren't perfectly symmetrical. Embrace the bend, embrace the T-shape, and embrace the awesome world of chemistry!