Okay, chemistry fans and curious cats, let's dive into a little secret. We're going to talk about bases. Not the "base" of a building, but the chemical kind. They're the ones that love to gobble up tiny, positively charged bits. Think of them as the ultimate huggers in the molecular world.
Now, you might think all bases are created equal. They all do their base-y thing, right? Well, not quite. It turns out there are different types of bases. It's like saying all dogs are dogs, but some are poodles and some are bulldogs. Different personalities, different approaches.
We've got our good old Lewis bases. These guys are super flexible. Their main gig is to share a pair of electrons. Imagine them offering a cozy little couch to a lonely proton. It's all about sharing and caring, in a chemical sense.
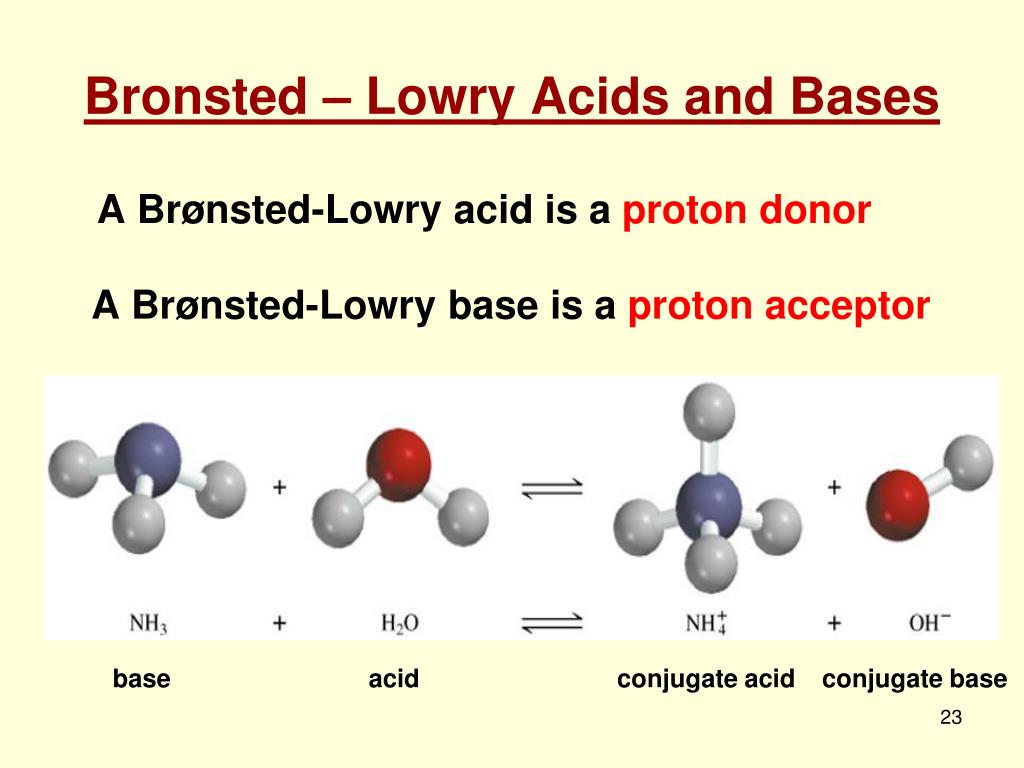
Then we have the Brønsted-Lowry bases. These are a bit more specific. They're all about accepting protons. A proton is just a hydrogen atom that's lost its electron. So, a Brønsted-Lowry base is basically a proton magnet. Zip! It snatches that proton right up.
Here's where things get interesting. The big question is: are all Lewis bases also Brønsted-Lowry bases? It's a juicy topic, a bit like asking if all squares are rectangles. Most people nod and say yes. But is that always true? Let's poke around a bit.
Think about water. We all know water, right? It's pretty cool stuff. Water can act as both a Lewis base and a Brønsted-Lowry base. It has those lone pairs of electrons to share, making it a Lewis base. And it can totally grab a proton from something else, becoming a Brønsted-Lowry base.
So, water is like a chameleon. It fits into both categories. That's a good sign, isn't it? It suggests a lot of overlap. If one common substance can do both, maybe many can.

Let's consider ammonia, NH₃. Ammonia has a nitrogen atom with a lovely pair of electrons just waiting to be shared. That makes it a classic Lewis base. When it meets an acid, like HCl, it readily accepts a proton from it. So, it's also a Brønsted-Lowry base!
It seems like the electron-sharing superpower of a Lewis base often comes with the proton-grabbing ability of a Brønsted-Lowry base. It’s like having two excellent skills for the price of one. Who wouldn't want that?
But here's the fun part, the little wrinkle in the fabric of chemistry. Are there any substances that are great Lewis bases but terrible Brønsted-Lowry bases? Or maybe even impossible to be a Brønsted-Lowry base?
Imagine a substance that's super electron-rich. It’s practically begging to share those electrons. That’s your ultimate Lewis base. But what if it can't accept a proton? What if it doesn't have anywhere to put it? Or what if it's too unstable when it does accept a proton?
This is where things get a little philosophical, or at least chemically quirky. Some molecules are fantastic at donating electron pairs. They are super nucleophilic, ready to attack anything with a positive charge. That's the Lewis base definition in full swing.

However, for a substance to be a Brønsted-Lowry base, it needs to be able to handle that incoming proton. Protons are, well, acidic. They can make things a bit wobbly. Some molecules just aren't built for that kind of party.
Consider something like boron trifluoride
, BF₃. Boron trifluoride has a boron atom that’s a bit electron-deficient. It’s like it’s always looking for a friend. It's a perfect example of a Lewis acid, meaning it wants electrons. But its partner, fluorine, is very electronegative. It pulls electrons towards itself.
Now, let's flip that idea. What about something that has a very strong hold on its own electrons and might not want to accept an extra proton? It might be a great electron pair donor but a terrible proton acceptor. It’s like someone who loves to lend you their tools but absolutely hates when you try to borrow their favorite hat.
Some experts would argue that technically, there are compounds that are excellent Lewis bases but not effective Brønsted-Lowry bases. These might be molecules where the electron pair is very localized or where accepting a proton would lead to an unstable arrangement.

Think about the humble halide ions, like chloride (Cl⁻). Chloride has those extra electrons and can share them, making it a Lewis base. It can also accept a proton to form HCl, so it’s a Brønsted-Lowry base too. See? Another overlap!
But what about really bulky molecules? Or molecules where the electron pair is tied up in some way? They might still be able to offer up electrons, but the proton might not be able to get close enough to accept. Or maybe accepting the proton just breaks the molecule apart.
It’s a bit like saying, Can all generous people also be excellent listeners?
Most generous people are probably pretty good listeners, right? But maybe some are so busy being generous they don’t quite hear what you’re saying. They're focused on the giving part.
The world of chemistry isn't always neat and tidy. It has its exceptions, its grey areas. And this question about Lewis and Brønsted-Lowry bases is one of those fun grey areas.
Most of the time, if something is a good Lewis base, it has the electron density to accept a proton. So, it will likely also be a Brønsted-Lowry base. It's a very common scenario.

However, the purest, most technical definition might allow for exceptions. Substances that are extremely stable with their electron pairs, or where accepting a proton leads to immediate destruction, might be the outliers.
So, while the answer is mostly yes, are all Lewis bases also Brønsted-Lowry bases? The true chemistry enthusiasts might give you a little smirk and say, Well, technically...
And that, my friends, is the fun of chemistry. It’s full of those little technicalities that make you scratch your head and smile.
It’s a fascinating thought. Are these two definitions so intertwined that they’re almost the same for most practical purposes? Or are there hidden gems of bases out there that only love to share electrons and have no interest in protons whatsoever?
It’s a playful debate. Most substances you’ll encounter in your everyday chemistry adventures will be both. But knowing there’s a possibility for a base to be a specialist – a pure electron-sharer – is what makes it all so exciting. It reminds us that even in science, there’s room for nuance and a little bit of mystery.
So next time you see a base, give it a knowing nod. It might be a multi-talented molecule, a true double threat in the chemical arena. Or it might be a purist, sticking to its electron-sharing roots. And that’s perfectly okay too!