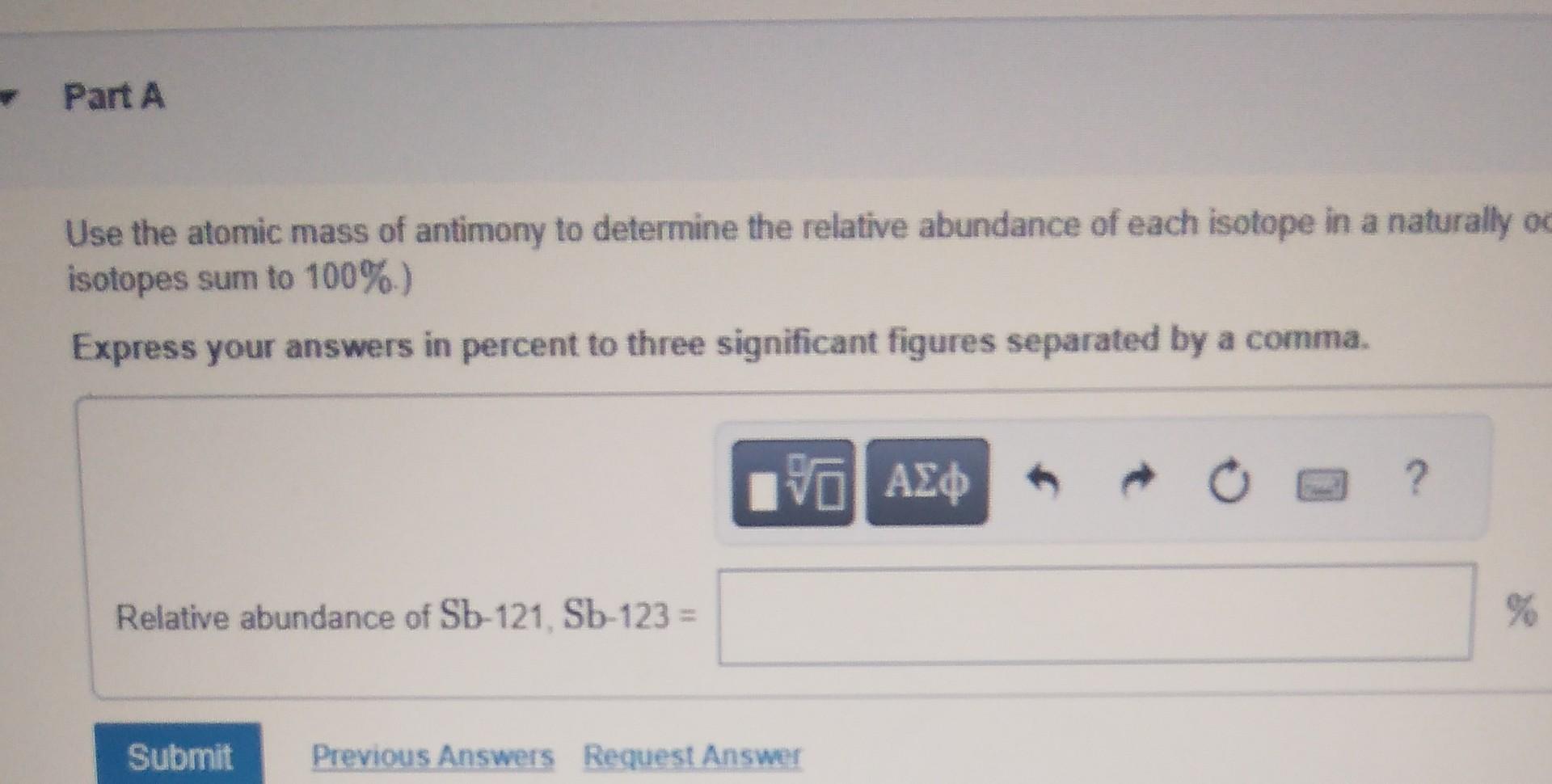
Have you ever thought about how the stuff around us, the very building blocks of everything, can be a little bit… peculiar? It’s a bit like discovering a secret handshake for a familiar object. Well, today we’re diving into the curious world of antimony, a fascinating element that, when you look closely, reveals a subtle, yet interesting, duality. It turns out that antimony, this often-overlooked metal, comes in not one, but two naturally occurring forms, or as scientists call them, isotopes. It’s a small detail, but understanding it can unlock a deeper appreciation for the complexity and wonder of the universe.
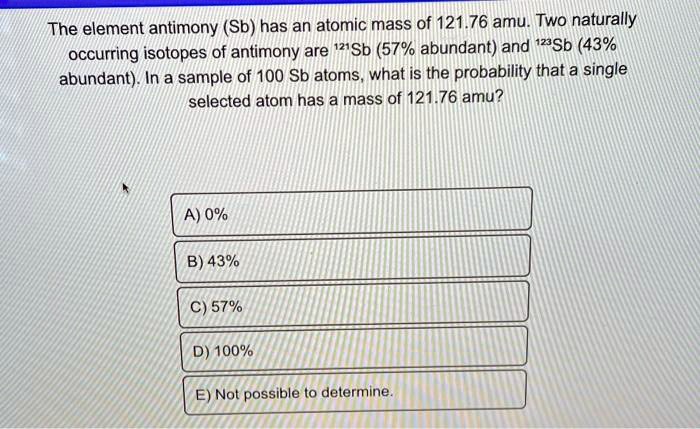
So, why should we care about antimony having two isotopes? It’s all about having a more complete picture. Think of it like a family portrait. You see the main subjects, but sometimes there are slightly different versions of that portrait – maybe one with a person squinting, another with them smiling. These are still the same people, but with subtle variations. For antimony, these variations are due to the number of neutrons in its atomic nucleus. The most common isotope, antimony-121, is what we encounter most often. But there’s also antimony-123, which is less abundant but just as much a part of antimony’s natural identity. This difference, though slight, can influence how antimony behaves in specific scientific contexts, making it a valuable tool for researchers.
The benefits of understanding isotopes like those of antimony are far-reaching, especially in fields like science education and industrial applications. For students, learning about isotopes is a stepping stone to understanding more complex concepts in chemistry and physics. It teaches them that elements aren't just monolithic entities but can have subtle internal variations. In daily life, though you might not see a label saying "contains antimony isotope X," these variations are crucial. For instance, antimony compounds are used as flame retardants in plastics and textiles, helping to keep us safer. Understanding which isotope is present can be important for precise manufacturing and safety regulations.
Educationally, exploring isotopes can be as simple as looking at a periodic table and noticing the atomic mass. It’s a hint that there’s more to the story than just a whole number. You can find educational videos online that explain isotopes in simple terms, often using analogies that make the concept click. In terms of daily life, while direct interaction with antimony isotopes is unlikely, you interact with their applications constantly. Think about the electronics you use – antimony compounds can be found in semiconductors. The next time you're near a fire extinguisher, remember that some fire-fighting chemicals might involve antimony's flame-retardant properties. It’s a quiet, behind-the-scenes contributor to our modern world.
For a bit of hands-on exploration, consider visiting a science museum. Many have exhibits that touch upon the fundamental nature of elements and their properties. You can also find readily available resources online, like interactive periodic tables that offer deeper dives into elements and their isotopes. Even a simple search for "what are isotopes?" will yield a wealth of informative and engaging content. It's a gentle invitation to look a little closer at the world around you and discover the subtle, fascinating stories that even the most common elements have to tell.