Hey there, future organic chemistry rockstar! So, you've dived headfirst into the wild and wonderful world of organic chemistry, huh? And you're staring down the barrel of acid-base practice problems? Don't sweat it! Think of it like learning to ride a bike – a little wobbly at first, maybe a few scraped knees (or maybe just a few confused brain cells), but eventually, you'll be cruising.
Seriously though, acid-base chemistry is like the alphabet of organic reactions. Get a solid grip on this, and everything else will start to make so much more sense. It’s all about who’s got the protons, who wants them, and who’s feeling generous with their electron pairs. Simple, right? (Okay, maybe not always simple, but we’ll get there together!).
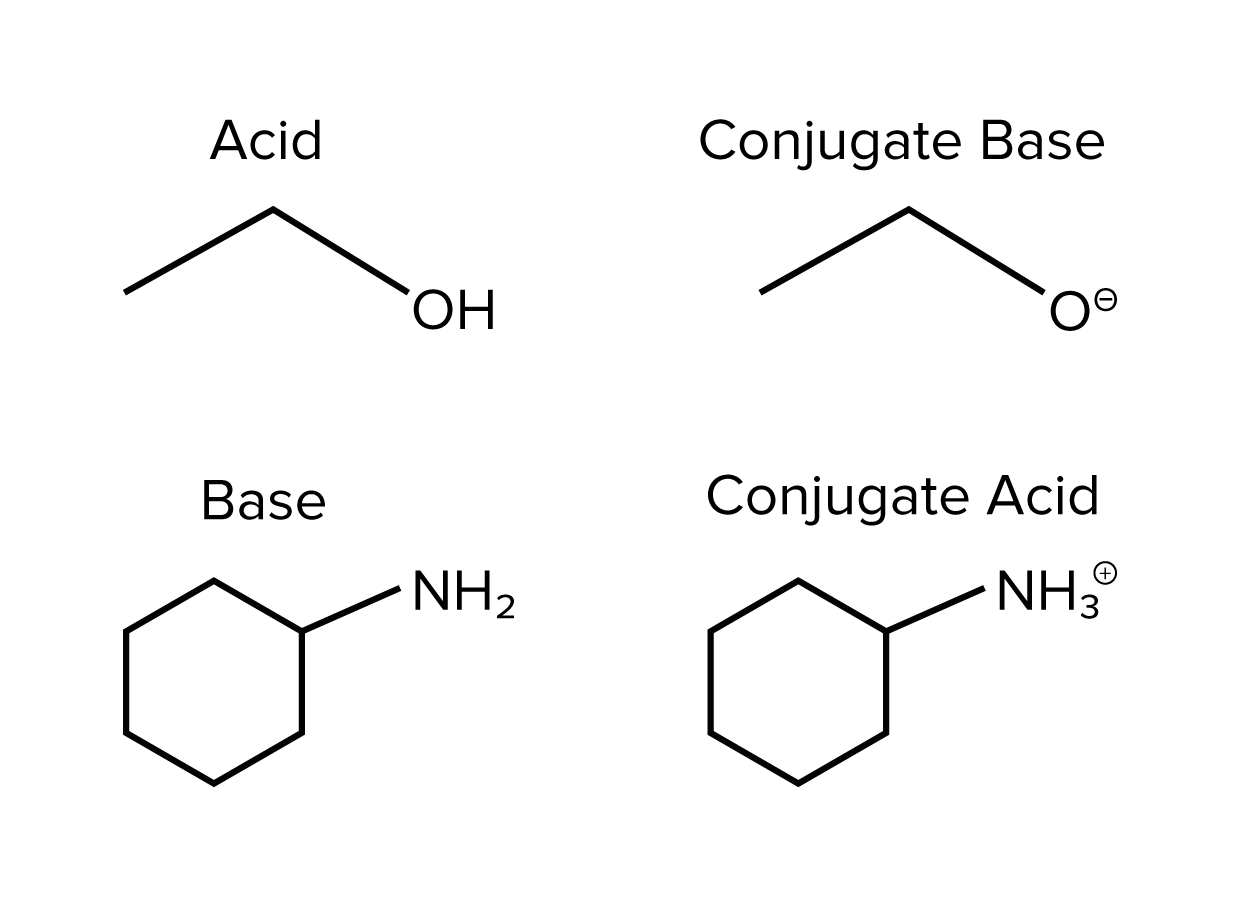
Let's ditch the stuffy textbook language for a minute. Forget about "Brønsted-Lowry definitions" and "conjugate pairs" being some kind of secret handshake. It's just about understanding that acids are proton donors (think of them as the generous ones, always handing out H+ ions) and bases are proton acceptors (they’re the receivers, happily scooping up those H+ ions). Easy peasy, lemon squeezy!
Now, why is this so darn important? Because so many organic reactions happen because of acids and bases. It’s the spark that ignites the whole process. If you don't know who's going to react with whom and why, you're basically trying to bake a cake without knowing what flour is. Disaster waiting to happen!
So, when you see an acid-base problem, the first thing you want to ask yourself is: "Who's going to give away a proton, and who's going to take it?" It's like a microscopic game of catch, but with a positively charged hydrogen atom. Wild!
The Magic Trio: Acids, Bases, and Their Friends
Let's break down the main players. You've got your acids, ready to shed a proton. Then you've got your bases, eager to grab one. Simple enough.
But then things get a little more interesting. When an acid loses its proton, what's left is called its conjugate base. Think of it as the acid’s "after-party" self. And when a base accepts a proton, it becomes its conjugate acid. It's like the base after a "proton-cation" (get it? Proton + elation! Okay, maybe my jokes need work, but you get the idea).
The real kicker? The strength of an acid or base determines how readily these proton transfers happen. We measure this strength using things like pKa values. Lower pKa? Stronger acid. Higher pKa? Weaker acid. It's kind of counterintuitive, like how the "worst" parking spots are often the most sought after because they're harder to get. Go figure!
So, when you're looking at a reaction, you'll often see an acid and a base on both sides. You'll have your reactants (the starting materials) and your products (what you end up with). The equilibrium, or where the reaction "prefers" to be, is often determined by the relative strengths of the acids and bases involved.
Common Organic Acids and Bases You'll Meet
You're going to bump into some regulars in these problems. Let's get acquainted:
Super Strong Acids (The Proton-Giving Machines)
- Hydrohalic acids: HCl, HBr, HI. These guys are notorious proton donors. They don't mess around.
- Sulfuric acid (H2SO4): Another heavy hitter. Handle with care!
- Nitric acid (HNO3): Don't let the pretty name fool you, it's a powerful acid.
Organic Acids (The Ones You'll See a Lot)
- Carboxylic acids: R-COOH. These are your bread and butter organic acids. Think acetic acid (vinegar!) or formic acid. They have that -COOH group which is a giveaway.
- Alcohols (sometimes): R-OH. While alcohols are usually weak acids, in the presence of a super strong base, they can donate their proton. It's like your quiet friend who can be surprisingly feisty when pushed.
- Phenols: Aromatic rings with an -OH group. These are generally more acidic than regular alcohols because the negative charge on the oxygen after losing the proton can be spread out across the ring (resonance, anyone? We'll get to that!).
Super Strong Bases (The Proton-Hogging Champions)
- Hydroxide ion (OH-): You'll see this one a lot.
- Alkoxides: RO-. These are the conjugate bases of alcohols. Think sodium methoxide (CH3O-Na+).
- Amide ion (NH2-): Very, very strong base. It's like the ultimate proton vacuum cleaner.
Organic Bases (The Ones That Like Protons)
- Amines: R3N. These guys have a lone pair of electrons on the nitrogen, making them prime candidates for grabbing a proton. Think of ammonia (NH3) and its organic cousins.
- Ethers (weakly): R-O-R. The lone pairs on the oxygen can sometimes act as a weak base.
The Art of Predicting Acid-Base Reactions: It's All About Equilibrium!
Here's the golden rule, the secret sauce, the thing you absolutely, positively, 100% MUST remember:
The equilibrium of an acid-base reaction favors the formation of the weaker acid and the weaker base.
Think about it: if you have a strong acid and a weak base, they'll react to form a weak conjugate acid and a strong conjugate base. That doesn't sound right, does it? The universe likes things to be stable. Strong acids are eager to get rid of their protons, and strong bases are eager to grab them. But once they've reacted, you've got a strong base and a strong acid on the product side. That's like having two magnets that repel each other – they're going to try and go back to their original, more stable forms.
So, the reaction will shift towards the side where the acid is weaker and the base is weaker. It's like a game of musical chairs, and the "stronger" players (acids and bases) get kicked out, leaving the "weaker" players to settle in. Everyone’s happier that way. Or at least, the molecule is!
Let's Practice! (The Fun Part… Ish)
Okay, theory is great, but practice makes perfect. Or at least, it makes you less likely to cry over your textbook. Let's walk through a typical problem.
Problem Example 1: Predicting the Major Products
Let's say you're given this reaction:
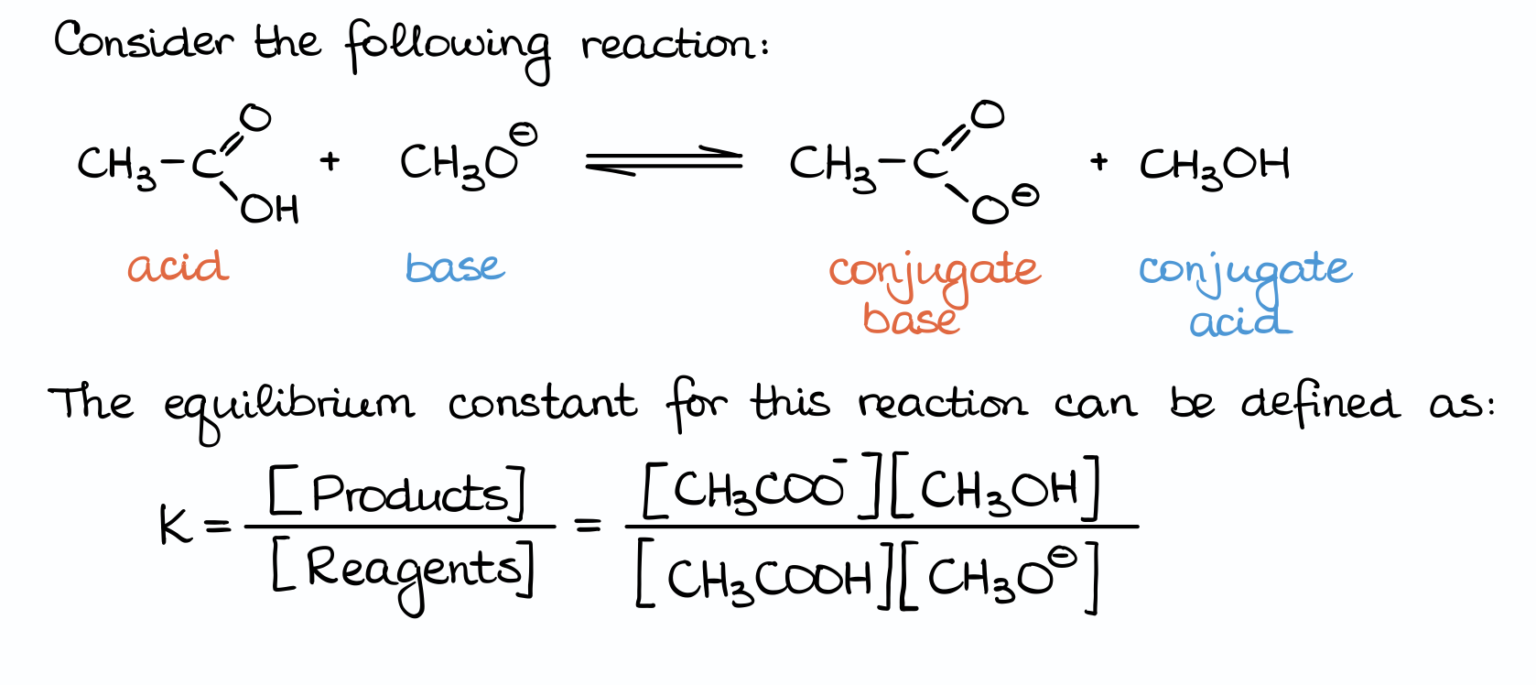
CH3COOH (Acetic Acid) + NaOH (Sodium Hydroxide) → ?
First, identify your players:
- CH3COOH: Acetic acid. This is a carboxylic acid, so it's an acid. Its pKa is around 4.76.
- NaOH: Sodium hydroxide. This dissociates into Na+ and OH-. The OH- is your base. It's a strong base.
Now, think about what happens when acetic acid loses its proton (the H+ from the -COOH group). You get CH3COO- (acetate ion). This is the conjugate base of acetic acid.
And what happens when hydroxide (OH-) accepts a proton? It becomes H2O (water). Water is a very weak acid, so in this context, it's acting as the conjugate acid.
So, the "potential" products are CH3COO- and H2O.
Now, apply the golden rule: equilibrium favors the weaker acid and weaker base.
We need to compare the acid strengths on both sides:
- Acid on the left: Acetic acid (pKa ≈ 4.76)
- Acid on the right: Water (pKa ≈ 15.7)
Which acid is weaker? Water (higher pKa = weaker acid). So, the equilibrium will lie to the right, favoring the formation of acetate ion and water.

The major product is:
CH3COO- (Acetate ion) + H2O (Water)
And the Na+ from NaOH just tags along as a spectator ion. So, the full reaction would be:
CH3COOH + NaOH → CH3COO-Na+ + H2O
See? Not so scary. You just have to play detective and figure out who's the proton donor, who's the acceptor, and who's the weaker one of the bunch.
Problem Example 2: Comparing Acid Strengths
Sometimes, you'll be asked to compare the acidity of two molecules. This is where your knowledge of functional groups and electron effects comes in handy.
Let's compare the acidity of ethanol (CH3CH2OH) and acetic acid (CH3COOH).

To do this, we look at their conjugate bases. The more stable the conjugate base, the stronger the original acid.
- Ethanol (CH3CH2OH): When it loses a proton, you get CH3CH2O- (ethoxide ion). The negative charge is localized on the oxygen atom.
- Acetic acid (CH3COOH): When it loses a proton, you get CH3COO- (acetate ion). Here, the negative charge is delocalized (spread out) across both the oxygen atoms of the carboxylate group through resonance.
Which conjugate base is more stable? The acetate ion (CH3COO-) is more stable because the negative charge is spread out. It’s like having a big pizza to share versus a tiny cookie all to yourself – everyone’s happier with the pizza!
Therefore, since the conjugate base of acetic acid is more stable, acetic acid is a stronger acid than ethanol.
This is why pKa values are your best friends. You can look them up if you're unsure!
Common Pitfalls to Avoid (So You Don't Facepalm)
We've all been there. Staring at a problem, convinced you've nailed it, only to realize you missed something obvious. Here are a few things to watch out for:
- Confusing acids and bases: Double-check which molecule is donating and which is accepting. It sounds silly, but in the heat of the moment, it happens!
- Forgetting about resonance: Resonance stabilization of a conjugate base is a HUGE deal. If you see a way to spread out that negative charge, do a little happy dance, because it means the acid is stronger.
- Ignoring inductive effects: Electronegative atoms (like halogens) near the site of the negative charge can pull electron density away, stabilizing the negative charge and making the acid stronger. Think of them as little "electron vacuums."
- Not comparing apples to apples: Make sure you're comparing the acid on the reactant side with the acid on the product side, and the base on the reactant side with the base on the product side.
- Thinking all alcohols are weak acids: While generally true, remember that strong bases can deprotonate them. Context is key!
Mastering the Art: Tips and Tricks
How do you get good at this? Practice, practice, practice! And some smart strategies:
- Draw it out: Always draw the structures and show the proton transfer. Seeing it visually can make a world of difference.
- Identify functional groups: Know your carboxylic acids, alcohols, amines, etc. Their behavior in acid-base reactions is usually predictable.
- Use a pKa table: Don't be afraid to consult your pKa table. It's there to help you! The more you use it, the more familiar you'll become with the relative strengths.
- Work through examples: Do as many practice problems as you can. Start with the easy ones and gradually work your way up.
- Explain it to someone else: Even if it's your pet goldfish, trying to explain a concept out loud helps solidify your understanding. (Your goldfish might not be the most attentive listener, but hey, it's the thought that counts!)
- Focus on the core principle: Equilibrium favors the formation of the weaker acid and weaker base. Keep this mantra in your head!
Acid-base chemistry might seem like a hurdle at first, but it's honestly one of the most fundamental and rewarding parts of organic chemistry. Once you crack the code, you'll feel this amazing sense of understanding. It's like unlocking a secret level in your favorite video game!
So, keep practicing, keep asking questions, and don't be afraid to make a few mistakes. Every problem you tackle is a step closer to mastering this essential skill. You've got this! Go forth and conquer those acid-base practice problems. You're going to do great, and soon you'll be able to predict reactions like a seasoned pro, with a smile and maybe even a bad chemistry pun or two. Happy studying!