Imagine a little piece of zinc metal. It looks pretty ordinary, right? Just a dull, silvery-gray chunk. But what if I told you this humble metal can put on a spectacular show when it meets the right partner? Get ready, because we're about to dive into a reaction that's truly something to behold!
We're talking about zinc metal. It’s got this hidden talent, a secret life waiting to be unleashed. And when it does, it's like a tiny fireworks display happening right before your eyes. It’s not just a science experiment; it’s a miniature marvel.
So, what’s this magical partner that makes our zinc metal perform? It’s something called a strong acid. Think of it as zinc’s ultimate hype-person. When these two get together, sparks – quite literally – start to fly!
When a sample of zinc metal reacts completely with a strong acid, it’s a moment of pure chemical excitement. The zinc doesn't just sit there; it transforms. It’s like watching a shy performer suddenly take center stage and belt out a show-stopping tune.
The whole process is surprisingly quick and visually dynamic. You see the zinc metal, minding its own business, and then BAM! The acid enters the picture, and the fun begins. It’s a visual feast for anyone who loves a bit of drama.
One of the most exciting things you'll notice is the bubbling. Lots and lots of bubbling! It’s as if the zinc is having a super energetic party. These tiny bubbles are actually a gas being produced, and they dance their way up from the metal.
This bubbling isn't just any old fizz. It's a sign of something significant happening. It means the zinc is actively breaking apart and changing into something new. It's chemistry in action, and it's happening at an impressive pace.
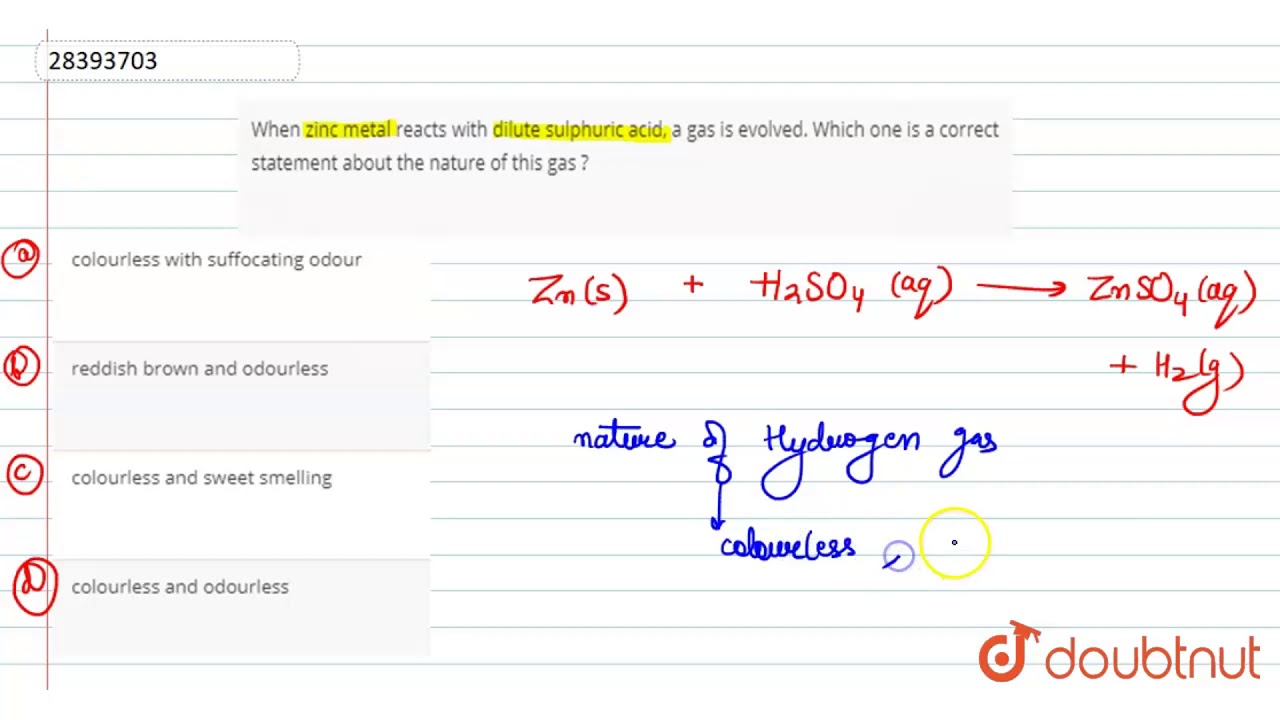
The gas produced during this reaction is called hydrogen gas. You might not see it, but it’s there, making its presence known through those enthusiastic bubbles. It’s the life of the party, this hydrogen gas!
And here’s where it gets even cooler: this hydrogen gas is flammable. That means if you introduce a tiny flame to it, you might get a little pop! It’s a delightful, albeit small, pyrotechnic demonstration. Safety first, of course, but the potential for a little flame trick is undeniably fun.
The reaction continues until all the zinc metal is used up. That’s what “reacts completely” means. Every single bit of the zinc gets involved in this energetic transformation. Nothing is left behind; it’s a full commitment!

This completeness is part of what makes it so satisfying to watch. You see the metal disappear, replaced by the lively dance of bubbles. It’s a clear indication that a significant chemical change has occurred, and there’s no going back.
The acid, the powerful catalyst, is also transformed. It's not just a passive observer; it's an active participant in this chemical ballet. It works diligently, helping to break down the zinc structure.
What’s left behind after the reaction is a solution. This isn't just plain water; it's a solution containing the products of the reaction. It's the lingering evidence of the zinc's grand performance.
Think of it like a chef preparing a meal. The raw ingredients (zinc and acid) go into the pot, and after some vigorous cooking (the reaction), you get a delicious dish (the new solution and gas). It’s a transformation you can see and even hear!
The speed of the reaction is also a key player in its entertainment value. It doesn't take ages to get going. Once the zinc and acid meet, the action is almost immediate. This quick onset keeps you engaged and wondering what will happen next.
It's the kind of science that makes you lean in and say, "Wow!" It’s accessible, it’s dramatic, and it’s a fantastic introduction to the world of chemical reactions.
You don't need to be a seasoned chemist to appreciate this. If you've ever been mesmerized by a bubbling potion or a colorful chemical display, you'll find this reaction utterly captivating.
The visual aspect is paramount here. The energetic fizzing, the potential for a tiny pop, and the visual disappearance of the solid metal all contribute to a highly engaging experience.

It’s a simple equation with a spectacular outcome. Zinc + Strong Acid = Exciting Reaction! It's a recipe for instant scientific fun.
Consider the humble origins of the zinc metal. It’s often overlooked, a solid, unassuming material. But its reaction with a strong acid reveals a hidden, energetic personality.
This transformation is a powerful reminder that even the most ordinary-looking substances can harbor extraordinary capabilities. It’s a lesson in looking beyond the surface.
The reaction also demonstrates the concept of reactivity. Some elements are naturally more inclined to react than others, and zinc is certainly eager when it comes to strong acids.
The rate at which the reaction occurs can also be influenced. Factors like the concentration of the acid and the surface area of the zinc can speed things up or slow them down, adding another layer of scientific intrigue.
Imagine placing a small piece of zinc in a beaker of acid. Within moments, you'll see the metal begin to dissolve, and the liquid will start to churn with tiny, energetic bubbles.
This isn't a slow, simmering process. It's a lively dance, a vigorous exchange between the metal and the acid. It’s like watching a tiny, controlled explosion of chemical energy.

The sound of the reaction can also add to the experience. While it's not a deafening roar, the constant effervescence creates a gentle, persistent fizzing noise that can be quite mesmerizing.
This auditory element, combined with the visual spectacle, creates a multi-sensory experience that makes the reaction even more memorable.
And then there’s the anticipation. As the zinc gets smaller and the bubbles become more vigorous, you know you’re witnessing something significant. You're seeing matter being rearranged at an atomic level.
It's a tangible demonstration of chemical principles that might otherwise seem abstract. You can see the zinc metal being consumed, and you can see the byproducts being formed.
The fact that the reaction goes to completion is key. It means you get to see the full effect of the chemical process. There’s a sense of closure and a clear endpoint to the energetic display.
What remains is a clear or slightly colored solution, depending on the specific acid used. It's the quiet after the storm, the aftermath of the energetic chemical event.
This simplicity makes it an ideal demonstration for educational purposes, but it's also just plain fun to observe for anyone with a curious mind.
So, if you ever get the chance to witness a sample of zinc metal reacting completely with a strong acid, seize it! It’s a small-scale spectacle that packs a big punch. It’s a fleeting moment of chemical magic, and it’s truly something special to behold.

You’ll walk away with a newfound appreciation for the hidden dynamism of everyday materials. It’s a reminder that science can be both fascinating and surprisingly entertaining, all thanks to a little bit of zinc and a lot of acidic energy!
It’s like watching a solid turn into a lively, bubbly gas right before your eyes!
The sheer activity is what makes it so engaging. It’s not a passive chemical change; it's a vigorous, dynamic process.
The visual cues are strong: the metal shrinking, the liquid churning, the bubbles rising relentlessly. These are all indicators of a powerful chemical transformation.
And the fact that it’s a complete reaction adds a sense of finality and satisfaction. You see the zinc go, and you witness the creation of new substances.
It's a perfect example of how chemistry can be both visually appealing and conceptually interesting. It’s a little showstopper in the world of chemical reactions.
So, next time you think about metals, remember the energetic potential of zinc. Its reaction with a strong acid is a testament to the hidden wonders of the elements around us.
It’s a reminder that even simple experiments can be incredibly rewarding and entertaining. Just a bit of zinc, a bit of acid, and a whole lot of chemistry happening!
![Chemical Properties of Metals [with Reaction Examples] - Teachoo](https://cdn.teachoo.com/large/9e28ee42-4e37-453c-8337-99e42863a4bf/reaction-of-zinc-with-steam---teachoo.png)

+%2B+Cu(SO4)(aq)+%EF%83%A8+Zn(SO4)(aq)+%2B+Cu(S).jpg)