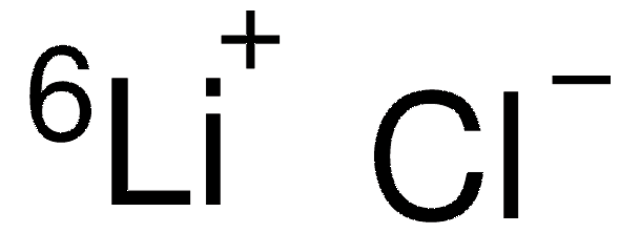Okay, so picture this: you're at a super fancy, maybe slightly bizarre, dinner party. The host, a quirky millionaire with a penchant for unusual appetizers, gestures to a small, shimmering bowl. "Do try my special blend," they chirp, their eyes twinkling with mischief. You lean in, expecting… I don't know, truffles? Saffron? And then, they whisper, "It's only lithium chloride!"
Your brain does a little stutter-step. Lithium? Isn't that for… you know… moods? And chloride? Sounds like something you'd find in a swimming pool. Your first thought might be, "Is this a prank? Am I about to get chemically altered into a placid, salt-loving blob?" Relax, grab a metaphorical napkin, and let's dive into this surprisingly fascinating, and dare I say, slightly edgy substance.
The Humble Beginnings of a Chemical Rock Star
So, what is this stuff, really? Lithium chloride (LiCl) is, at its core, a salt. Like the salt you sprinkle on your fries, but instead of sodium and chlorine, it’s lithium and chlorine doing the tango. Think of it as the introvert cousin of table salt – a bit more reserved, a bit more… energetic in its own way.
Now, lithium itself is pretty darn cool. It's the lightest metal on Earth. Seriously, you could probably float a brick made of pure lithium on water. And it’s so reactive, it’s like a toddler at a candy store – always eager to jump into things. Chlorine, on the other hand, is a bit of a drama queen, always wanting to pair up with something.
When these two get together, they form lithium chloride. It’s a white, crystalline solid, and in its pure form, it’s surprisingly… well, normal-looking. No neon glow, no bubbling spontaneously (usually). It’s the quiet kid in the chemical classroom that, upon closer inspection, has some seriously wild secrets.
It's Not Just for Your Mood Swings (But Kind Of)
Okay, let's address the elephant in the room, or rather, the lithium in the lithium chloride. Yes, lithium is famous for its role in treating bipolar disorder. It's like a chemical thermostat for your brain, helping to smooth out the extreme highs and lows. So, does eating a bowl of lithium chloride make you instantly zen? Probably not. And please, for the love of all that is chemically stable, do not try eating it.

The way lithium chloride is used medically is in highly controlled doses, specifically as a mood stabilizer. It’s not like popping a vitamin. It’s a powerful medication that requires careful monitoring. So, while it shares the name, the context is entirely different from that fancy dinner party appetizer.
Think of it this way: a Ferrari and a go-kart both have wheels and an engine, but you wouldn't use a go-kart to win the Indy 500. Similarly, while both medical lithium and lithium chloride contain lithium, their applications and dosages are worlds apart.
Surprising Superpowers: Beyond the Medicine Cabinet
But here’s where things get really interesting. Lithium chloride isn't just a one-trick pony. It's got some sneaky, under-the-radar talents that make it a bit of a chemical workhorse.
First off, it's a desiccant. That's a fancy word for something that loves to suck moisture out of the air. Imagine a super-powered sponge for humidity. This makes it incredibly useful in drying out gases and other substances. So, while you might be trying to add moisture to your skin, lithium chloride is out there, valiantly fighting off dampness.
It's also got a pretty low melting point for a salt. We’re talking around 614 degrees Celsius (1137 Fahrenheit). That might sound hot to you and me, but in the grand scheme of things, it's practically a lukewarm bath compared to some other compounds. This property makes it handy in certain industrial processes where controlled melting is key.
The Air Conditioner's Secret Weapon?
And here's a mind-blower: lithium chloride plays a role in some advanced air conditioning systems. Yep, the thing that keeps you from melting into a puddle of sweat during a heatwave might be using this unassuming salt. How? Well, its hygroscopic nature comes into play again. It can absorb moisture from the air, effectively dehumidifying it, which makes the cooling process more efficient. So, next time you're enjoying that icy blast of air, give a silent nod to the humble lithium chloride.

It's like the unsung hero of your comfort, working behind the scenes while you're blissfully unaware. It's not flashy, it doesn't have a catchy jingle, but it's out there, making your life a little bit cooler, one water molecule at a time.
When Things Get a Little Too Salty (Or Not Salty Enough)
Now, as with most things in life, it’s all about balance. While pure lithium chloride is interesting, its true magic often happens when it's mixed with something else. But the prompt specifically said "a mixture consisting only of lithium chloride." This is where the fun lies! A pure substance, in and of itself, can be fascinating. It's like appreciating a single, perfectly cut diamond.
However, in the real world, you’ll rarely find it in isolation. Its uses often involve it being dissolved in water (making a lithium chloride solution) or interacting with other chemicals. But for the sake of our thought experiment, let's marvel at the pure compound. It’s the solo artist, the prima donna of its own chemical stage.

But let's not forget, even a pure substance has properties that can be… dramatic. If you were to, hypothetically, spill a significant amount of pure lithium chloride, it’s quite soluble in water. This means it would dissolve rather enthusiastically. And while it's not as overtly dangerous as, say, a vat of concentrated acid, it's still a chemical that needs to be handled with respect. It can irritate skin and eyes, and ingesting it in large quantities would be a very bad idea, potentially leading to all sorts of unpleasantness, from nausea to more serious effects.
The Takeaway: It's Not Just a Flavoring
So, the next time you hear about lithium chloride, don't just think "mood stabilizer" or "swimming pool." Think about the unassuming white crystals that can help control air conditioners, dry out gases, and have a surprisingly low melting point for a salt. It’s a testament to how even seemingly simple chemical compounds can have a complex and vital role in our world.
It’s a little bit of a rebel, a little bit of a helper, and a whole lot of interesting. And while I still wouldn't recommend it as a dinner party snack, I can certainly appreciate its quiet, chemical competence. It's the reliable friend who never makes a fuss but always gets the job done, whether that job is keeping you calm, keeping you cool, or just being a really, really good desiccant. Pretty neat, huh?